Properties of Water & Macromolecules

Water and Solutions
MAIN IDEA: The properties of water make it well suited to help maintain homeostasis in an organism.
Do Now:
Name one way our bodies use water to maintain homeostasis
https://www.youtube.com/watch?v=HVT3Y
3_gHGg
Properties of Water
Water is polar; universal solvent
Water is cohesive.
Water is adhesive.
Water is less dense as a solid.
What is a solvent?
What happens when you take kool-aid powder and pour it into a cup of water?
Discuss with someone close to you; what is the solute? What is the solvent?
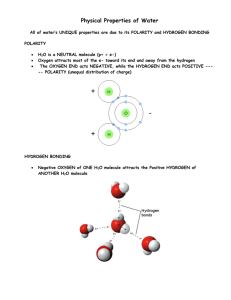
Polarity
polar molecules - molecules that have an unequal distribution of charges
Polarity is the property of having two opposite poles.
hydrogen bond - weak interaction involving a hydrogen atom and a fluorine, oxygen, or nitrogen atom; happens b/c of polarity; strong type of van der Waals force
Cohesive
Molecules of water – attracted to each other b/c of H-bonds
Causes surface tension
Allows water to form droplets
Allows insects to rest on water’s surface
Adhesive
Water forms Hbonds with molecules on other surfaces
Allows water to travel up stems of plants
(capillary action)
Question…
Is a meniscus an example of cohesion or adhesion?
Discuss with someone close to you
Ice is less dense than water
As water cools to 4 °C, it becomes more dense.
When it freezes, it is less dense than liquid water.
Nutrients in bodies of water mix.
Animals live under frozen surface of bodies of water in winter.
Question…
What would happen in winter to animals that live in lakes if water was more dense as a solid? Think
Discuss with someone next to you
What is a mixture?
Substances combine, but do not change chemically
Can be separated easily
No chemical change occurs
Ex: tea, saltwater, salad dressing, bag of candy
Types of Mixtures
Homogeneous – mixture looks the same throughout
Ex: salt water, soda
Heterogeneous – mixture is different throughout
Ex: salad, salad dressing
Solutions
Homogenous mixture
Molecules of one substance mix evenly w/those of another (dissolves)
2 parts:
Solute – substance that is dissolved
Solvent – substance that does the dissolving
Name solute & solvent in salt water
Concentrations of solutions
Dilute – less solute
Concentrated – more solute
Saturated – solution has as much solute as it can hold.
Colloids
2 substances that don’t mix evenly
Stay mixed
Usually thicker than most liquids
If light is shined through, it scatters –
Tyndall effect
Ex: fog, milk, jell-o, cream
Tyndall Effect
Suspension
2 substance that don’t mix evenly
Do not stay mixed
Particles – heavy and settle
Ex: blood, flour and water, aerosols, ice cream
Compare and contrast heterogenous and homogenous mixtures:
Acids and Bases
Acids
release hydrogen ions (H+) when dissolved in water
Ex: stomach acid, vinegar, citrus fruit
Bases
release hydroxide ions(OH-) when dissolved in water
Ex: ammonia, soap, blood
pH and buffers
pH - measure of concentration of H+ in a solution
Acidic solutions - pH values lower than 7.
Basic solutions – pH values above 7.
pH of 7 = neutral
Buffer – mixtures that react with acids or bases to keep the pH in a neutral range
Water
Water pH = 7 (neutral)
Water splits into H + and OH -
Equal amounts of each
Salts
Formed when acid and base react
Ex: NaOH + HCl NaCl + HOH
base acid salt water
Needed to control many life processes
6.4: The Building
Blocks of Life
MAIN IDEA: Organisms are made up of carbon-based molecules.
Organic Chemistry
Carbon is a component of almost all biological molecules.
4 electrons in outer energy level, so 4 electrons to share in 4 covalent bonds
Carbon
Carbon makes covalent bonds with other elements, like hydrogen, oxygen, and nitrogen (CHON)
Carbon molecules – can be straight chains, branched chains, or rings
Macromolecules
Carbon atoms join to form carbon molecules.
Macromolecules - large molecules formed by joining smaller organic molecules together.
Polymers - molecules made from repeating units of identical or nearly identical compounds linked together by a series of covalent bonds.
Each link - monomer
Monomer
Polymer
4 Main Organic Compounds
Carbohydrates, lipids, proteins, and nucleic acids
All needed for proper cell function and structure
Carbohydrates
Carbon, hydrogen, oxygen in ratio of
1:2:1
Ex: glucose = C
6
H
12
O
6
Reduces to 1:2:1
(CH
2
O) n
Key energy source in most foods
Provides structural support in cells
Ex: sugars and starches
Monosaccharides
Values of n ranging from three to seven are called simple sugars, or monosaccharides.
(saccharide = sugar)
Building blocks of carbohydrates
Ex: glucose, fructose
Longer Carbohydrates
2 monosaccharides = disaccharide
More than 2 = polysaccharide
Lipids
Made mostly of carbon and hydrogen
Nonpolar, so not soluble, or mostly insoluble, in water
Types of Lipids
Phospholipids – make cell membranes
Steroids/sterols – ex: cholesterol – in animal cell membranes
Pigments – ex: chlorophyll
Fats, oils, and waxes
Fats and Oils
Store a LOT of energy
Mostly C-H bonds, which have a lot of energy
Structure usually – 3 fatty acids bonded to glycerol (called triglyceride)
Fat if solid at room temp; oil if liquid at room temp
Fatty acids
Saturated fats = all C’s bonded to at least
2 H’s; single bonds between carbons; most animal fats (butter, lard, grease)
Unsaturated = some double bonds between carbons (1 = monounsaturated; more than 1 = polyunsaturated); liquid at room temp (oils); healthier to eat
Hydrogenated = H added to unsaturated fats to improve texture
Proteins
Made of amino acids – small carbon compounds made of CHONS
(carbon, hydrogen, oxygen, nitrogen, sometimes sulfur)
20 different amino acids
Amino Acid Structure
Contain a central carbon atom
One of carbon’s bond is to hydrogen
The other three bonds are with an amino group ( –NH2), a carboxyl group (–COOH), and a variable group ( –R).
Bond between 2 amino acids = peptide bond
Complexity of Protein Structure
Primary structure – number and order of amino acids joined together
Chain of amino acids = polypeptide
Secondary structure – chain folds into a 3-D shape: helix or pleat
Protein Structure
What are proteins for?
Enzymes – promote chemical reactions
Structure of organisms: collagen (skin, ligaments, tendons..), bone, hair, muscles
Provide antibodies and hormones
Allow muscle contractions, blood clots
Hemoglobin (carries oxygen in blood)
Nucleic Acids
Store and transmit genetic information
Ex: DNA and RNA
Made of nucleotides – repeating units made of PCHON
Sugar, base, phosphate group
DNA and RNA- both have 4 types of nucleotides
DNA – double helix (2 chains, spiral); genetic material
RNA – one strand, makes proteins
Fill in the blanks:
KOH + HBr
HCl +
LiOH + HBr
+ H
2
O
KCl + H
2
O
+
______ + ______ RbF + H
2
O
ANSWERS!
KOH + HBr KBr + H
2
O
HCl + KOH KCl + H
2
O
LiOH + HBr LiBr + H
2
O
HF + RbOH RbF + H
2
O
DO NOW
Draw the atomic structure and the
Lewis structure for magnesium (Mg).
Is magnesium stable? If not, what should it do to become stable?
Fill in the blanks:
HF + LiOH +
Energy, Work, and Order
Energy = ability to do work or cause change
Potential = stored energy
Kinetic = energy of motion
For work to occur, potential energy must be converted to kinetic energy
Energy in Cells
Energy is used for many things in cells:
Moving substances
Building new molecules
Growth
Reproduction
Establishing and maintaining order
Establishing/Maintaining Order
Atoms/molecules arranged in specialized order
Organization allows cells and systems to function properly
EX: cells organized to tissues, organized into organs, and finally body systems.
Takes ENERGY to maintain organization
Energy and Order
Cells, atoms, etc, WANT to be in a state of disorder
Without energy, systems become simple/disorganized (entropy)
Continual input of energy keeps a state of order
Organisms = highly organized systems b/c of constant energy input
Where is the energy from?
Energy in Reactions
Synthesis rxns use energy
Often, they react very slowly
Need catalysts
Catalyst = substance that promotes chem rxns but is not affected/used up
Enzyme = specialized catalyst in organisms.
Photosynthesis
Process by which autotrophs (producers) like plants (and some bacteria and green algae) make their own food
Uses water, carbon dioxide, and energy from sunlight to make glucose (sugar)
Takes place if organism has chlorophyll
(green pigment)
Usually happens in leaves
Photosynthesis cont.
Synthesis reaction
Requires energy in form of sunlight
Photosynthesis cont’d sunlight
Water + carbon dioxide oxygen + glucose
6H
2
O + 6CO
2 sunlight
6O
2
+ C
6
H
12
O
6
Glucose = sugar; stored as sugar to give plants energy, and starch to give consumers energy
Photosynthesis cont’d
Plants get water from roots, transported to leaves by xylem
Stomata – “doorways” into leaves for gases
CO
2 in and O
2 out
Gases cannot pass through waxy part of leaf
Sunlight captured in chloroplasts in cells, which produce chlorophyll (pigmentabsorbs light)
Stomata
Chloroplast
Respiration
Glucose energy
Decomposition reaction
Occurs in mitochondria
ALL organisms go through respirationsome aerobic, some anaerobic
Mitochondria: “the powerhouse”
Respiration cont’d
Glucose + oxygen carbon + water + energy dioxide
C
6
H
12
O
6
+ 6O
2
6CO
2
+ 6H
2
O + energy
Energy Molecules
Energy stored in ATP as chemical energy
ATP: adenosine triphosphate
ATP ADP
ADP: andenosine diphosphate
Remains when ATP is used
Cycle replaces ATP supply
3 Steps of Respiration
1.
2.
3.
Glycolysis
Krebs Cycle
Electron Transport System
Glycolysis (pg. 397)
Glyco = sugar
Lysis = to split apart
Glucose split in half
Net 2 ATP
Krebs cycle (pg. 399)
3-carbon molecules disassembled
Carbon dioxide released
Energy released – gain of 2 ATP (4 total so far)
Hydrogen also released electron transport
Electron Transport Chain (pg. 400)
Hydrogen’s electrons transferred from carrier to carrier, releasing energy
“Caught” by oxygen – makes water
(released)
34 ATP gain (38 total)
44% energy from glucose ATP
Only 25% of gas in car usable energy!
Oxygen/Carbon Dioxide Cycle
Oxygen is a waste product of photosynthesis, but is used in respiration.
Carbon dioxide is a waste product of respiration, but is used in photosynthesis.
Two Types of Chemical Rxns
Synthesis
Compounds are made
Energy is required/used
Ex: Making glucose
(photosynthesis)
Decomposition
Compounds are broken down
Energy is released
Ex: breaking down food
(digestion)
Synthesis or Decomposition?
Na + Cl NaCl
NaCl Na + Cl
Synthesis or Decomposition?
Na + Cl NaCl synthesis
NaCl Na + Cl decomposition
Ionization Reactions
Ionic bonds can separate in solution
Ex: salt in water separates to Na + and
Cl ions in the water
Compounds NOT made of ions can undergo ionization
Ex: water – not made of ions, but very few water molecules separate into ions
Important for many life functions
H
2
O H + + OH -
H
2
O H + + OH -
H + = hydrogen ion (H lost an e-, and now it’s only a proton)
OH = hydroxide ion (it has the e- lost by the hydrogen ion)
Ionized water: hydrogen ions = hydroxide ions
When hydrogen ions ≠ hydroxide ions, the solution is an acid or a base