Inorganic Compounds
advertisement

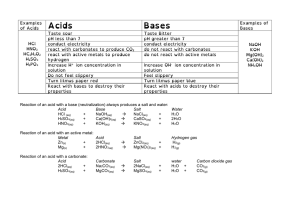
Inorganic Compounds Water 1. The most abundant inorganic compound found in living organisms is . 2. About 65-75% of the body weight of humans is composed of water. What percentage of the following are composed of water? Percent Water Red Blood Cells Muscle Tissue Blood Plasma 3. How long can you live without water? 4. Several chemical and physical properties of water explain its vital role in living organisms. a. Water is a universal . b. The versatility of water as a solvent is due to its bonds. Because of its polar nature, having positive and negative end of the molecule, other molecules, especially compounds, readily dissolve in water. c. Water participates in catabolic chemical reactions such as , where water is added to large organic nutrient molecules to break them down. d. Water absorbs and releases very slowly, thus moderating the effects on body temperature of fluctuations in external temperatures. e. The primary way humans lose heat and regulate their body temperature is by cooling when evaporates from the surface of the skin. Acids and Bases 1. Acids hydrogen ions into solution, where as bases hydrogen ions from solution. 2. pH is the measure of concentration in a particular substance. 3. Neutral solutions have a pH of . Acids have a pH range from to and bases have a pH range from to . 4. On the diagram below, create a scale from 0 to 14. Indicate the sections of higher H+ ion concentration and OH- concentration. Indicate the acidic, basic, and neutral portions of the scale. Then place the following substances on the scale: pure water, blood, urine, stomach acid, household ammonia, vinegar, oven cleaner, household bleach, tomatoes, grapes, ocean water, eggs, saliva, and milk. 5. Buffers are substances that help maintain a pH within They will hydrogen ions if body fluid is too basic and hydrogen ions if body fluid is too acidic. limits (pH 7.35 to pH 7.45). Salt 1. Salts are 2. Salts are 3. Electrolytes have the ability to compounds that do not contain H+ or OH-. , which can carry electrical currents in the body. in water.