Alcohols
advertisement

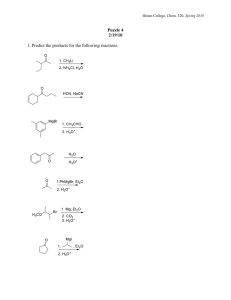
Alcohols Biological Activity Nomenclature Preparation Reactions Some Alcohols CH3CH2OH HO OH OH CHCH2NH2 CHCHNHCH3 CH3 ethanol HO adrenaline (epinephrine) OH H HOCH2CHCH2OH glycerol H HO H cholesterol pseudephedrine Alcohols are Found in Many Natural Products HO N CH3 O H HO Morphine most abundant of opium's alkaloids Paralytic Shellfish Poisoning NH2 O O H HN H N N NH N A possible chemical warfare agent roughly 1000 times more toxic than saran gas or cyanide N H OH The toxin blocks entry of sodium OH required by cells to make "action potentials" Saxitoxin (STX) LD 50 = 2 g/kg OH O O OH OH HO OH O H2N OH OH OH OH HO PALYTOXIN LD 50 = 0.15 g/kg OH OH OH OH OH OH O HO O N N H H HO OH OH OH OH OH O OH OH OH OH HO OH O O O OH HO OH OH OH OH HO OH OH OH OH OH Ethanol: the Beverage Ethanol is a central nervous system depressant - depresses brain areas responsible for judgement (thus the illusion of stimulation) alcohol dehydrogenase CH 3CH 2OH ethanol NAD + O CH 3CH + NADH + H acetaldehyde LD 50 = 1.9 g/Kg NAD enz. + CH 3CO 2H + NADH + H acetic acid + + Methanol: Not a Beverage CH3OH methanol ADH NAD + O + HCH + NADH + H formaldehyde LD 50 = 0.07 g/Kg Alcohol Nomenclature OH 3 3-heptanol 6 2 5 5-methyl-6-hepten-2-ol OH 2 OH 1 1 3 CH3 CH3 3,3-dimethylcyclohexanol OH CH3 5 CH3 5,5-dimethylcyclohex-2-enol Nomenclature OH OH (E) 3-methyl-3-penten-2-ol (S) 2-hexanol OH OH trans 3-isopropylcyclopentanol H OH (R) 2-butyl-1,4-butanediol (R) 2-butylbutane-1,4-diol Oxidation levels of oxygen- halogen- and nitrogencontaining molecules CH2=CH2 CH3CH3 [O] CH3CH2OH HC [O] CH CH3CH=O [O] CH3CO2H CH3CH2Cl CH 3CHCl2 CH3CCl3 CH3CH2NH2 CH3CH=NH CH3CN Oxidation Reduction Acidity of Alcohols • Due to the electronegativity of the O atoms, alcohols are slightly acidic (pKa 16-18). • The anion dervived by the deprotonation of an alcohol is the alkoxide. • Alcohols also react with Na/K (same as water does) to give the alkoxide. CH3CH2OH + Na CH3CH2O Na + 1/2 H2 Withdrawing Groups Enhance Acidity F H F C OH F > more acidic alcohol CH3OH CH3CH2OH CF3CH2OH (CH3)3COH (CF3)3COH Why? F C OH H H > H C OH H less acidic pKa 15.54 16.00 12.43 18.00 5.4 Withdrawing Groups Enhance Acidity CF3 CF3 CF3 C OH + NaHCO3 CF3 CF3 alcohol CH3OH CH3CH2OH CF3CH2OH (CH3)3COH (CF3)3COH C O Na + H2CO3 CF3 pKa 15.54 16.00 12.43 18.00 5.4 A similar case for phenols Physical Properties b.p. oC D sol. in H2O CH3CH2CH3 -42 0.08 i CH3OCH3 -25 1.3 ss CH3CH2OH 78 1.7 vs Intermolecular H-Bonding O H H H O H O H O associated liquid intermolecular H bonding O H H H O Preparation of Alcohols • • • • Reduction of ketones and aldehydes Reduction of esters and carboxylic acids Hydration of Alkenes Nucleophilic addition – Grignard reaction – Acetylide addition • Substitution • Epoxide opening NaBH4 Reduction O R 1) NaBH4, ethanol R' 2) H3O + H H OH R R' H3O H R O R' + Some Examples O OH 1) NaBH 4, ether 2) H3O O CH + " CH2OH Two Alcohol Products Form in Lab O H axial approach NaBH4 (CH3)3C H O Na (CH3)3C trans O Na O NaBH4 (CH3)3C H (CH3)3C H equatorial approach cis LiAlH4 Reduction a Stronger Reducing Agent OH O 1) LiAlH 4, THF 2) H3O + LiAlH 4 will reduce: o ketones to 2 alcohols o aldehydes to 1 alcohols o carboxylic acids and esters to 1 alcohols LiAlH4 is a much stronger reducing agent O 1) LiAlH 4 2) H3O OH + + CH3OH O 1) NaBH4 2) H3O + no reaction NaBH4 is More Selective O O 1) NaBH4 OH 2) H3O OH + O OH OH 1) LiAlH 4 2) H3O + OH Oxymercuration Hydration Markovnikov 1) Hg(OAc) 2 in THF/H2O 2) NaBH4 OH H Hydroboration Hydration Anti-Markovnikov 3 1) BH3-THF 2) H2O2, NaOH H OH 3 Base Catalyzed Ring-Opening of Epoxides Acid Catalyzed Ring-Opening Aqueous and in Alcohol Nucleophilic addition to Carbonyl Compounds Acetylides O H O C CH3 CH3CH2C CH3 CH3 CH3CH2C C CH3 C C H3O + OH CH3 CH3CH2C C C CH3 Organometallic Chemistry Grignard Reaction CH3 Br + Mg "CH3 MgBr " excellent nucleophile very strong base CH3 MgBr Grignard Reagent Grignard Reagents React With Ketones to form tertiary alcohols O CH3 1) CH3MgBr in ether 2) H3O HO + MgBrOH + o a 3 alcohol H3O MgBrO CH3 CH3 + Grignard Reagents React With Aldehydes to form secondary alcohols O MgBr in ether 1) H OH + 2) H3O H Grignard Reagents React With Formaldehyde to form primary alcohols CH2CH2O MgBr CH2CH2OH H3O + O C H H formaldehyde CH2 MgBr CH2Br Mg, ether, Grignard Reagents react (twice) with Esters to form 3o Alcohols O OH C C CH 3 CH3 OCH3 1) 2 CH3MgBr 2) H3O + CH3 O C OCH 3 CH3 2nd eq. 1) CH3MgBr + 2) H3O O C CH3 ketone (more reactive than ester) Grignard Summary H H R MgX + + C O H3O workup R H formaldehyde R MgX R' + O R' H3O workup R H aldehyde R MgX R' + C R'' ketone C OH H R' + O OH H + C C H3O workup R C R'' OH Grignard Summary R O H3O workup R' MgX + epoxide R'' R R' 2 R MgX + OH + R' + C O RO ester H3O workup R C OH R + ROH Grignard Reagents are exceptionally strong bases H2O CH3OH CH3CH2CH2MgBr + CH3CO2H HC CH CH3NH2 CH3CH2CH3 Synthesis OH ? Retrosynthetic Analysis OH ? Br MgBr 4-Step Synthesis OH 1) HCHO + 2) H3O Br 2, h Br Mg in ether MgBr Synthesize Using Only 1,2, or 3-Carbon Reagents OH HC CH Retrosynthesis + OH O MgBr HC Mg Br CH HBr CH3X CH3X reduce Reactions of Alcohols Oxidation R-X, Ether, and Ester Preparation Protection of Alcohols Synthesis The Logic of Mechanisms Alcohols are Synthetically Versatile Oxidation - Reduction Oxidation of 2o Alcohols with Cr(VI) Mechanism Na2Cr2O7 + H2O + 2 H2SO4 O OH + HO o 2 alcohol O Cr OCrO3H OH O Chromic Acid (Cr VI) CrO3H H 2 H2CrO 4 + 2 NaHSO4 + H2O Chromate ester O OH2 + H3O + HCrO3 ketone (Cr IV) Oxidation of 1o Alcohols PCC oxidizes 1o Alcohols to Aldehydes CrO3Cl N PCC H pyridinium chlorochromate Oxidation of 1o Alcohols to Aldehydes: PCC Oxidation Summary CH2CO2H Na2Cr2O7 H2SO4 CH2CH2OH NH CrO3Cl OO DMSO, ClCCCl (CH3CH2)3N, in CH 2Cl2 CH2CHO CH2CHO Reduction Summary CH2CO2H 1) LiAlH 4 2) H3O + CH2CH2OH 1) NaBH 4 2) H3O CH2CHO + or H2, Raney Ni CH2CHO Conversion of Alcohol into a Leaving Group • Form Tosylate (p-TsCl, pyridine) • Use strong acid (H3O+) • Convert to Alkyl Halide (HX, SOCl2, PBr3) Formation of p-Toluenesulfonate Esters Best to use p-TsCl with pyridine CH3 OH CH3 O CH3 + ClS O p-toluenesulfonyl chloride OS N pyridine reacts with HCl as it forms O O N H Cl CH3 Reactions of Tosylates: Reduction, Substitution, Elimination CH3 OH CH3 O + ClS O CH3 O OS pyr: CH3 O 1) LiAl H4 KI NaOCH3 CH3 CH3 CH3 I H + LiOTs Alcohols to Alkyl Halides OH HX (HCl or HBr) X rapid S N1 + HOH o 3 alcohol OH HX moderate S N1 o 2 alcohol X + HOH 1o and 2o Alcohols: best to use SOCl2, PBr3, or P/I2 All are SN2 Reactions SOCl2 pyridine OH PBr 3 P, I2 (in situ prep. of PI3) Cl Br I Dehydration of Alcohols – E1 OH H H2SO4 (aq) cat. + H2O H regenerated H O HSO 4 or H2O H -H2O H Use the ff building blocks to synthesize the target compound. O CH3OH CH3OH OCH3 OH 1) Br 2, h PCC or + Na2Cr2O7, H 2) Mg P/I2 or CH3I MgBr H3O + O 1) Na 2) CH3I + OH Provide a sequence of steps OH Br OH HO 2 Approaches OH Br HO OH PCC KOH, DMSO in CH 2Cl2 O Br OH 1) CH3MgBr in ether H 2) H3O + Br Alternate Approach OH Br HO OH ClSi(CH 3)3 pyridine Br 1) CH3MgBr in ether 2) H3O OSi(CH3)3 + KOH, DMSO (SN2) HO OSi(CH3)3 PCC in CH 2Cl2 H OSi(CH3)3 O Problem Set: Road Map Problem Br A MgBr B O 1) CH3CH2CH + 2) H3O C Na2Cr2O7 H2SO4 D 1) CH3MgBr + 2) H3O E The Williamson Ether synthesis uses an alkoxide and alkyl halide… • Ethers (R-O-R) • SN2 reaction between R-X and R-O- The Williamson Ether synthesis uses an alkoxide and alkyl halide… • SN2 reaction between R-X and R-O• WE NEED TO CONSIDER STERIC HINDERANCE. This might lead to E2! Backside attack is not favorable! Methoxide is also a very strong base. The Williamson Ether synthesis uses an alkoxide and alkyl halide… • Practice: The Williamson Ether synthesis uses an alkoxide and alkyl halide… • Practice: Reactions of Phenols • Conversion to ethers • Electrophilic aromatic substitution • Oxidation to quinones Conversion to Ethers Electrophilic Aromatic Substitution Oxidation: Quinones Reactions of Ethers Ethers are quite UNREACTIVE. For most ethers, NO REACTION with mild acids, bases, halogens, nucleophiles, etc. Possible reactions include: • Acidic ether cleavage • Epoxide ring opening Cleavage by acids Sulfur analogs of alcohols and ethers are called thiols and sulfides. Thiols can be made via SH- and RX, and can react via Williamson mechanism Disulfides are formed via oxidation. Reduction to thiols can occur as well. http://b.vimeocdn.com/ts/147/230/147230470_640.jpg http://delight.spslinfotechpvtl.netdnacdn.com/media/catalog/product/cache/1/image/650x650/9df78eab3 3525d08d6e5fb8d27136e95/r/e/rebonding.jpg Disulfides are formed via oxidation. Reduction to thiols can occur as well. For rebonding: (1) Thioglycolate (acid-like) to convert disulfide bonds in hair protein to thiolates (2) Hydrogen peroxide to oxidize the thiolates back to disulfides. (3) Reforming the disulfides helps realign amino acids and make hair straight http://delight.spslinfotechpvtl.netdnacdn.com/media/catalog/product/cache/1/image/650x650/9df78eab3 3525d08d6e5fb8d27136e95/r/e/rebonding.jpg Disulfides are formed via oxidation. Reduction to thiols can occur as well. “antioxidant” because it protects your cells from oxidative degradation. http://b.vimeocdn.com/ts/147/230/147230470_640.jpg