GHW#4-Questions
advertisement

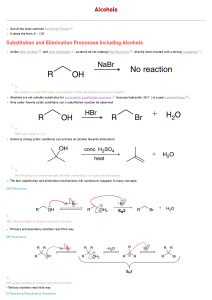
Chapter 14 and GHW#4 Questions Introduction Alcohols, Phenols, and Ethers: 1. Bonding Characteristics of Oxygen Atoms in Organic Compounds Structural Characteristics of Alcohols 1. Identify the Alcohols, Phenols, and Ethers from the following and give their common and/or IUPAC names. 1. Identify the Alcohols, Phenols, and Ethers from the following and give their common and/or IUPAC names. 2. Circle the constitutional isomers of 1- hexanol among following alcohols. 3. Classify each of the following as 1', 2', or 3' alcohols. a) d) b) c) Chemical Reactions of Alcohols 1) Combustion: Alcohol are very flammable and the combustion products are carbon dioxide and water. 2) Substitution Reactions: OH group is replaced by a another group such as halogen. a) Elimination Reactions: Intramolecular dehydration : Water is removed from a alcohol molecule and a alkene is produced. ( H2SO4 at 180) Intermolecular dehydration : Water is removed from two alcohol molecules and an ether is produced. ( H2SO4 at 140) Williamson Ether synthesis. 4a. Complete the following reactions of alcohol. a) Dehydration: 4b) What is Zaitsev Rule? Zaitsev’s rule (or the opposite of Markovnikov’s rule) Dehydration of an alcohol can result in more than one alkene product, because hydrogen loss can occur from either of the neighboring carbon atoms. Hydrogen is removed from the carbon with lowest hydrogen atoms ( poor get poorer) 5) Complete the following reactions of alcohol oxidation: 6) Draw condensed formula of each of the following: a. 3-Hexanol b. 1,2,3-Pentanetriol c. 2-Methyl-2-pentanol d. Cyclohexanol e. 3,4-dimethyl-3-heptanol 6) Draw the alkene products of the dehydration of the following alcohols: a. 3-Hexanol b. 1,2,3-Pentanetriol c. 2-Methyl-2-pentanol d. Cyclohexanol e. 3,4-Dimethyl-3-heptanol 7) Draw the alkene products of the dehydration of the following alcohols: a)2-Pentanol: b)3-Methyl-1-pentanol: c) 2-Butanol: d)4-Chloro-2-pentanol: e)1-Propanol: 8) Give the oxidation products of the following alcohols. If no reaction occurs, write N.R.