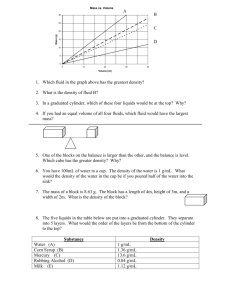
Properties of Matter Unit Review: Answer on a separate sheet of
advertisement

Properties of Matter Unit Review: Answer on a separate sheet of paper. Name________________________ 1. What defines matter? Anything that has both mass and volume is matter. 2. What are the four basic properties of matter? Mass, volume, weight and density. 3. What is inertia? It is an object’s tendency to resist a change in motion. Emphasis on the “change” in motion. Inertia can only be experienced when accelerating (changing motion.) 4. How can you measure inertia? Mass is the measure of inertia. So you could use a any balance that measures mass such as a triple beam balance. 5. How can you tell if one object has more inertia than another? One way would be to mass both objects on a balance. Whichever one has more mass, also has the greater inertia. Another way to would be to accelerate both objects. Whichever one takes more force to speed up, would have more inertia. 6. What is mass? Mass is a measure of how much matter something is. Mass is also the measure of inertia. 7. What does mass measure? Mass is the measure of inertia. 8. What is weight? Weight is the measure of the force of gravity. The equation for weight is F g = m ag 9. What is the weight of a 5 kg mass? Fg = m ag F = 5 * 10 = 50 [N] 10. Compare the weight of an object on the Earth to the moon. An object would weigh less on the moon because there is less gravity on the moon compared to the Earth. 11. What is volume? Volume is a measure of the amount of space that an object takes up. 12. How do you calculate the volume of a box, and of a cylinder? Volume for a box is equal to length times width times height or V = l w h. Volume of a cylinder is equal pi times the radius squared times the height or V = π r2 h. 13. How do you find the volume of an object with a graduated cylinder? Put some water in the graduated cylinder and measure its volume. Then put the object under the water line in the graduated cylinder. Then measure the new volume in the graduated cylinder. Lastly, subtract the original volume from the new volume for the volume of the object. 14. What is density? Density is the ration of mass to volume, or a measure of how much mass is in a given amount of space. For example a density of 5 [g/cm3] represents a mass of 5 [g] in a space of 1 [cm3] 15. Find the density of a 50 g mass that has a volume of 5 ml. D = m / V D = 50 / 5 D = 10 [g/ml] 16. What classifies as a fluid? Give an example of each type. A fluid is any liquid or gas. Or it is any substance with a changing shape. Liquid water and gaseous air are both fluids. 17. What has more mass a kilogram of something or a kilogram of something else? Since the [kg] is the unit for mass, than 1 [kg] of something has the same mass as 1 [kg] of something else. 18. What has the larger volume, a kilogram of deflated balloons, or a kilogram of inflated balloons? The inflated balloons take up more space than the deflated balloons. 19. Of scales and balances name one that truly measures mass, and another one that truly measures weight. A triple beam balance truly measures mass, and electronic balance truly measures weight. Also a mass balance measures mass, and produce scale measures weight. 20. How can you use a graduated cylinder and an electronic balance to find the density of an object? Use the electronic balance to measure the mass of the object, then use the graduated cylinder to measure the volume of the object. Lastly use the density equation D = m / V to solve for the density. 21. Where is it easier to lift of box of beans? On earth or on the moon? It is easier to lift a box of beans on the moon because it weighs less there due to the lower amount of gravity. 22. Where is it easier to throw a box of beans? On earth or on the moon? It’s just as hard to throw the box of beans on the moon as it is on the Earth because the box has same mass or the same resistance to a change in motion in both locations. 23. Describe the steps for using a triple beam balance. 1) move all the sliding mass back to the zero position. 2) Make sure the balance is zeroed out. 3) Place the object on the balance pan. 4) move the large sliding mass over one step at a time until it tips the scale, then back up one step. 5) Repeat step 4 with the medium mass. 6) Move the small sliding mass over until the scale balances. 7) Add up the values of the three sliding masses for the mass of the object. 24. Describe the steps for using an electronic balance. 1) Press the power button to turn the electronic balance on. 2) Press the tare/zero button to zero out the balance. 3) put the object on the electronic balance pan. 4) read the mass of the object. 25. A mass balance is used to find the mass of a golf ball. It takes 3 dimes (2 g each), 2 pennies (2.5 g each), and 4 nickels (5 g each) to balance out the golf ball. Find the mass of the golf ball. The mass of the golf ball is (3 * 2) + (2 * 2.5) + (4 * 5) = 6 + 5 + 20 = 31 [g] 26. A triple beam balance is used to measure the mass of an action figure. When balanced the 100 g mass is in the 1 position, the 10 g mass is in the 8 position, and the 1 g mass is in the 2 position. What is the mass of the action figure? The mass of the action figure is (100 * 1) + (10 * 8) + (1 * 2) = 100 + 80 + 2 = 182 [g] 27. A graduated cylinder reads 22 ml before putting in a paper clip. Then it reads 26 ml. What is the volume of the paper clip? V = 26 -22 V = 4 [ml] 28. Chapstick has a radius of 1 cm and a height of 4 cm. What is the volume of the chapstick? V = π r2 h V = π 12 4 V = 12.6 [cm3] 29. A matchbox has a length of 8 cm, a width of 5 cm and a height of 3 cm. What is the volume? V = l w h V= 8 * 5 * 3 V = 120 [cm3] 30. The mass of a frog is 150 g and its volume is 3 cm3. What is the density of the frog? D = m / V D = 150 / 3 D = 50 [g / cm3] 31. What are the density rules for what an object will do when surrounded by a fluid? 1) If the object is less dense than the fluid it will rise and float on the surface. 2) If the object is more dense than the fluid it sinks to the bottom. 3) If the object is equally dense as the fluid it suspends in the middle of the fluid. 32. Why does ice float? Ice is less dense than water therefore it rises in the water and floats at the surface. 33. How far should you safely stay away for an iceberg that measures 250 ft wide above the surface? Since only 10% of the iceberg is visible above the surface of the water the general rule is to stay 10x farther away than what you can see. So in this case 10 * 250 = 2,500 ft. So stay 2,500 ft away to safely pass the iceberg. 34. What happens to the density of an object if the volume increases, and b) decreases? If the volume increases the density decreases. If the volume decreases the density increases. 35. What happens to the density of an object if the mass increases, and b) decreases? If the mass increases the density increases. If the mass decreases than the density decreases. 36. List and define the four states of matter? Solid: constant shape, constant volume. Liquid: changing shape, constant volume. Gas: changing shape, changes volume, neutral charge. Plasma: changing shape, changing volume, all charged ( + or -) particles. 37. List 2 examples of each state of matter. Solid: ice, brick. Liquid: water, lava. Gas: Air, clouds. Plasma: lightning, the Sun. 38. What do water and steam have in common? What do steam and ice have in common? What do ice and water have in common? What do water, steam and ice have in common? Water and steam both have a changing shape. Steam and ice are both less dense than water. Ice and water both have a constant volume. Water, steam and ice are all H2O. 39. What are the characteristics of a physical change? Give two examples. A physical change does not change the identity of the substance is and is usually reversible. Chopping wood. Pouring water in a sports bottle. 40. What are the characteristics of a chemical change? Give two examples. Chemical changes do change the identity of the substance, and are difficult or impossible to reverse. Baking cookies, burning marshmallows. 41. What happens when a marshmallow is caught on fire? How can you prove this is a chemical change? The outside of the marshmallow burns, and turns back. There is a new substance created, carbon. There is nothing that can be done to turn the carbon layer back into marshmallow. 42. What happens when a tea bag is placed in hot water? How was it proven that making tea is physical change? The tea mixes with the water to create a solution of water and tea. If the tea water solution is boiled the water is given off as steam and the tea remains behind. Making tea is considered a physical change because the look and taste change but the tea and water never form a new substance, they just get mixed together. 43. Define Chemistry. What is a Chemist? Chemistry is the study of matter and its changes. A chemisty is a person that studies matter and its changes. 44. What is an endothermic reaction? What happens to the temperature of surrounding area? A endothermic reaction is process in which heat is absorbed by the reaction. This makes the surrounding area colder since heat was removed from it. 45. What is an exothermic reaction? What happens to the temperature of the surrounding area? An exothermic reaction is a process in which heat is released by the reaction. This makes the surrounding area warmer as heat was added to it. 46. What is a molecule? List two examples of molecules. What happens if you break down a molecule into smaller parts? A molecule is the smartest part of the substance that is still that substance. H 2O is a water molecule. NaCl is a salt molecule. If you break a molecule down into smaller pieces you end up with a different substance. Break H2O down into Hydrogen and Oxygen, neither of which is water. 47. What is oxidation? Is oxidation a physical or chemical change? Oxidation is a chemical change in which oxygen reacts with iron to iron oxide or rust. 48. What is photosynthesis? Describe the chemical reaction of photosynthesis in words. Photosynthesis is the process by which plants use energy from the sunlight to create food. Photosynthesis is when six molecules of water plus six molecules of carbon dioxide react with sunlight to form one molecule of sugar and six molecules of oxygen. 49. What is tarnish? How does it form? Tarnish is a chemical reaction where silver reacts with oxygen to form silver oxide or the black coating that forms on silver. 50. What makes baking different from burning? Baking is controlled heat (proper temperature and time). Burning is excessive or too much heat (too much temperature and/or time.) Both processes are chemical changes.