Unit_3_assignment_-_..
advertisement

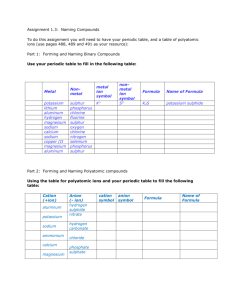
CHEMISTRY 11 Unit 3 Assignment – Naming Compounds Name: /76 Date: PART A Complete the following chart: ( /60) Name or write the formula & state whether the substance is molecular (M), Ionic (I), Acid (A), Element (E). Type M, I, A or E 1. Chemical Name sodium chloride CaCO3 2. 3. 4. nickel (III) bisulfate sodium hydroxide CaO 5. MgSO4•7H2O 6. 7. 8. 9. 10. Chemical Formula carbon dioxide acetic acid carbon calcium sulfate Na2SiO3 11. Ca(HCO3)2 12. 13. 14. magnesium hydroxide potassium chloride CHEMISTRY 11 Unit 3 Assignment – Naming Compounds 15. sodium thiosulphate pentahydrate 16. sodium hypochlorite Na2CO3 17. HCl (aq) 18. 19. potassium nitrate CuSO4•5H2O 20. Mg 21. 22. magnesium oxide KI 23. H2SO4 (aq) 24. 25. 26. silicon monocarbide iron Ca(OH)2 27. 28. potassium hydroxide S 8 29. 30. NH4Cl CHEMISTRY 11 Unit 3 Assignment – Naming Compounds PART B Answer the following questions: ( /16) 1. List the diatomic elements & how can you recall them?(4) 2. What are the elements that can be multiatomic, show them in this form.(2) 3. What is the difference between an Atom, Molecule and an Ion?(3) 4. What artificially separates the metals & non-metals? (1) 5. What does the chemical formula of an acid usually start with? (1) CHEMISTRY 11 Unit 3 Assignment – Naming Compounds 6. What does the chemical formula of a base usually end in? (1) 7. What two types of particles form an Ionic compound? (1) 8. What type of particles combine to form molecular/covalent compounds? (1) 9. Some compounds contain H2O in their structure what are these compounds called? (1) 10. Name a polyatomic ion that has a positive charge. (1)