2.222 Practice Problems 2003
advertisement

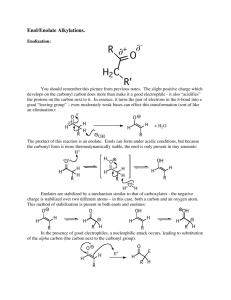
2.222 Practice Problems 2003 Set #3 In addition to those suggested in the list of general problems from Fox and Whitesell, do Supplementary Problems 15.15, 15.17, 15.21, 15.22, 15.23, 15.24 and 15.25 (the one we worked part-way in the last class). Answers to these are available via the same link you used for other Fox and Whitesell Supplementary problems; click here. I realize that most of you are too busy studying just now to pursue general interest items, but maybe after the exam you will have some time. I highly recommend reading Chapter 23 in the textbook (Molecular Basis for Drug Action) for anyone who has been stimulated by 2.222. You might also like to read the article mentioned in the “Chemical Perspectives” box on page 751 of the textbook, Tetrahedron, 46, 7599 (1990), which is available in the Science Library. This describes the classic synthesis of Chlorophyll A by R.B. Woodward and co-workers, which was accomplished in the late 1950’s but did not get published until 1990, long after Woodward’s death (a brilliant scientist, but stupid about his health. He chain-smoked himself into an early grave.) Here are three pretty challenging mechanism problems for you to practise on. 1) Give a reasonable mechanism for the following reaction. What kind of reaction is this? OH heat O OH O 2) The following reaction was used in the total synthesis of dihydrocostunolide, a member of the “germacrodiene” family of compounds that is secreted by various species of pine trees as a defense against insect damage. The reaction comes from Ziegler, F.E.; Piwinski, J.J. J. Am. Chem. Soc. 1979, 101, 1611-1612. several steps heat O (iPr)3Si O O A B O Si(iPr)3 O O dihydrocostunolide Provide a mechanism for the transformation of A into B. There are actually TWO sequential reactions involved here, both of which we have seen. Note that the (iPr)3Si group (tri-isopropylsilyl) is a protecting group, and one of the “several steps” later in the synthesis involves its removal. 3) Two equivalents of ethyl acetoacetate react with one equivalent of formaldehyde in the presence of a base to form the cyclohexenone product shown. This process involves two aldol steps and a Michael addition, although not necessarily in that order. Write out a detailed stepwise mechanism for this transformation. OEt O O O 2 OEt + O H NaOEt H EtOH OEt O O 2.222 Practice Problems 2003 Answers to Set #3 Answers to the suggested Supplementary Problems from Chapter 15 are available here. The three mechanism problems were deliberately chosen to be tough – if you understand these, you are probably doing OK. 1) OH O heat OH O This is a great example of the kind of problem that has given the class trouble in the past. It is not obvious what kind of reaction this is at first glance, and you really must use a systematic approach or you will end up going off on a tangent. 1) WHAT is happening? a) A 4-membered ring is opening up. b) An alkene group has disappeared. c) Two tertiary alcohols have been replaced by two ketones. d) Notice that the product is a 6-carbon chain with CH3 groups at positions 3 and 4, and oxygen at positions 2 and 5. So is the starting material! 1 3 O OH 4 5 3 1 2 5 2 4 O OH 6 6 Now we can see that the 2-5 bond is broken in this reaction. What kind of reactions do we know of that could simultaneously break a sigma bond and a pi bond in a ring, just by heating? ELECTROCYCLIC REARRANGEMENTS! 2) HOW is this occurring? Now we have some idea of how we might push the arrows. Let's give it a try to see whether it makes any sense. Note that we are not certain yet, but we at least have a working hypothesis. It is essential that we push the arrows correctly if we want to test that hypothesis, but we don't need to see exactly what the result will be before we start out. 1 1 OH 3 2 5 4 6 OH 3 2 5 4 OH OH 6 Remember that in an electrocyclic rearrangement, the number of pi and sigma bonds changes by one each - and this is accomplished by the "merry-go-round" of electrons within the ring. The product is interesting because it contains 2 enol groups. We know that enols are just the unstable tautomers of ketones, so we are on the right track. Let's redraw the structure in an extended geometry and tautomerize the first enol using a trace of acid. OH 1 2 3 H+ 5 6 4 OH 1 2 OH 3 +O And then, the same thing again on the other enol: H OH +O + 5 3 3 H 1 1 6 4 O 2 1 - H+ 6 6 O O O 5 4 H 5 4 3 2 6 4 OH 2 - H+ 5 1 2 3 5 6 4 O 2) heat O (iPr)3Si O O O (iPr)3Si A O O (iPr)3Si Now, it may not be obvious, but this final structure is identical to our target, compound B. 7 8 8 6 7 9 5 10 11 O 1 (iPr)3Si 4 12 O 6 5 4 3 9 10 11 3 2 2 12 1 O O Si(iPr)3 B This is just a Cope rearrangement followed by an Ester Enolate Claisen rearrangement. 3) OEt O O O 2 OEt + O NaOEt EtOH H H OEt O O We know that this reaction involves two Aldols and a Michael. This means that we need to look for bonds in the product that could be formed by reactions of this type, in order to see WHAT we must accomplish in our mechanism. OEt O This CH2 comes from formaldehyde. CH2 OEt The 2 acetoacetate fragments are coloured red and blue. We can see 3 bonds that must be formed in our mechanism. O O We know that the central CH2 group of acetoacetate is the most acidic site, and so our mechanism must begin here, with an Aldol involving formaldehyde. O O OEt O O OEt OEt -OEt H O O O O OEt H H HOEt O O H HO H EtOO O H OEt EtOH EtO- OEt O O O EtO OEt EtOOEt O O OEt O O O OEt O OEt O O OEt O O O O O O O O H OEt HO EtOH EtO O EtO O PRODUCT!