Photoelectron Spectroscopy
advertisement

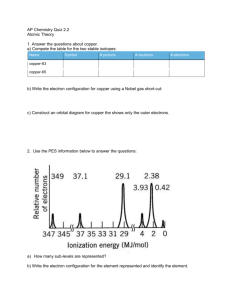
Are you interested in up to 10 test bonus points? Go to either web site (or both) and write a minimum of a 5 question worksheet that applies to either or both of these websites. Make sure to include all pertinent information and email to kbaugher@lphs.org. by Friday 10-31 before 5:00 pm. Links to Photoelectric Effect simulators http://phet.colorado.edu/en/simulation/photoelectric http://www.chem.arizona.edu/chemt/Flash/photoelectron. html Photoelectron Spectroscopy (PES) Unit 6 AP CHEM What is PES? Photoelectron spectroscopy (PES) is a technique used for determining the ionization potentials of molecules. Underneath the banner of PES are two separate techniques for quantitative and qualitative measurements. They are ultraviolet photoeclectron spectroscopy (UPS) and X-ray photoelectron spectroscopy (XPS). UPS focuses on ionization of valence electrons while XPS is able to go a step further and ionize core electrons and pry them away. • When a sample surface is irradiated with photons of energy hν, electrons are emitted from the sample surface. The figure below shows the essence of this photoemission process. This process was first reported by H.Hertz in 1887. The excitation source was a UV lamp. Nowadays, the excitation source can be X-ray or synchrotron radiation. The Photoelectric Effect • Albert Einstein considered electromagnetic energy to be bundled into little packets called photons. •Energy of photon = E = hv • Where, h = Planck constant ( 6.626 x 10-34 J s ) • v = frequency (Hz) of the radiation •Photons of light hit surface electrons and transfer their energy hv = B.E. + K.E. •The energized electrons overcome their attraction (to the nucleus) and escape from the surface (binding energy) • Photoelectron spectroscopy detects the kinetic energy of the electron escaped from the surface. PES and AP Chemistry • Our Studies will be mainly to look at data (spectrums) and try to explain the findings. The photoelectron spectra above show the • For example: energy required to remove a 1s electron from a nitrogen atom and from an oxygen atom. Which of the following statements best accounts for the peak in the upper spectrum being to the right of the peak in the lower spectrum? (A) Nitrogen atoms have a half-filled p subshell. (B) There are more electron-electron repulsions in oxygen atoms than in nitrogen atoms. (C) Electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p subshell of nitrogen atoms. (D) Nitrogen atoms have a smaller nuclear charge than oxygen atoms. More about PES The diagram opposite shows an energy level diagram for sodium with approximate binding energies for the core levels. If we are using Mg Kα ( hν = 1253.6 eV ) radiation ... ... at what kinetic energy will the Na 1s photoelectron peak be observed ? (the 1s peak is that resulting from photoionization of the 1s level) 1253.6 eV – 1072 eV = 181.6 eV ... at what kinetic energy will the Na 2s and 2p photoelectron peaks be observed ? 1253.6 eV – 64 eV = 1189.6 eV 1253.6 eV – 31 eV = 1222.6 eV Between 1190 and 1223 eV. Finally • The physics behind the PES technique is an application of the photoelectric effect. For solids, photoelectrons can escape only from a depth on the order of nanometers, so that it is the surface layer which is analyzed. • Because of the high frequency of the light, and the substantial charge and energy of emitted electrons, photoemission is one of the most sensitive and accurate techniques for measuring the energies and shapes of electronic states and molecular and atomic orbitals. Photoemission is also among the most sensitive methods of detecting substances in trace concentrations, provided the sample is compatible with ultra-high vacuum and the analyte can be distinguished from background. • Typical PES (UPS) instruments use helium gas sources of UV light, with photon energy up to 52 eV (corresponding to wavelength 23.7 nm). The photoelectrons that actually escaped into the vacuum are collected which results in a spectrum of electron intensity as a function of the measured kinetic energy. Because binding energy values are more readily applied and understood, the kinetic energy values, which are source dependent, are converted into binding energy values, which are source independent. This is achieved by applying Einstein's relation. Explain the data collected for the 1s electron in carbon, oxygen, nitrogen and fluorine. Explain the location of the peaks for sulfur and silicon for the 2p electron. What would you explain about this spectrum? Photoelectron Spectroscopy Conclusions • It uses quantum mechanics to interpret spectroscopic data and extract information on atomic structure from such data. In particular, lowresolution PES of atoms provides direct evidence for the shell model. • Light consists of photons, each of which has energy E = hv, where h is Planck’s constant and v is the frequency of the light. In the photoelectric effect, incident light ejects electrons from a material. This requires the photon to have sufficient energy to eject the electron. • PES determines the energy needed to eject electrons from the material. Measurement of these energies provides a method to deduce the shell structure of an atom. The intensity of the photoelectron signal at a given energy is a measure of the number of electrons in that energy level. • The electronic structure of atoms with multiple electrons can be inferred from evidence provided by PES. For instance, both electrons in He are identical, and they are both roughly the same distance from the nucleus as in H, while there are two shells of electrons in Li, and the outermost electron is further from the nucleus than in H.