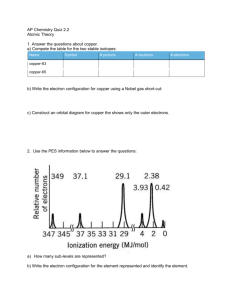
Photoelectronic Spectroscopy (PES) AP Chemistry Quantifiable
advertisement

Photoelectronic Spectroscopy (PES) AP Chemistry • Quantifiable application of the photoelectric effect • If light shining on a material (especially a metal) has sufficient energy it is able to eject electrons from that material (Each photon: E=h, see Ch. 6) • Photoelectron spectroscopy (PES) determines the amount of energy needed to do so • Measurement of these energies deduce the structure of the atom • Intensity of the signal number of electrons at that energy level • Note: energy is measured in eV or MJ/mole electrons Example of PES Spectrum Generated by the simulation at; http://www.chem.arizona.edu/chemt/Flash/photoelectron.html PES Example Questions 1. If the question flagged Mg as the element in question: a. Which peak in the spectrum represents electrons that are closest to the nucleus? b. What is the relevance of the relative height of the peaks at 5.31 and 9.07 eV? c. Why is there such a large difference in energy between the peaks at 0.74, 5.31 and 9.07 eV and the peak at 126 eV? 1 2. If the question did not flag Mg as the element in question: a. Assuming that the PES data shows ALL the electrons present in the atom, identify the element. b. Still applying the assumption above, which specific electrons are associated with the peak at 126 eV? c. What does a relatively low value of energy tell you about the relative position of the electrons within any atom? 3. Mg is found in group 2 and period 3 of the periodic table. Use this information to answer the following questions: a. Predict the full electronic configuration of Mg using s,p,d,f notation. b. Use your answer in (a.) to predict the relative heights of the four peaks found in the PES plot for Mg. Recommended Supplemental PES Material: Coulomb’s Law and AP Chem: Paul Andersen http://www.bozemanscience.com/ap-chem-004-coulombs-law/ PES in more depth: UC-Davis Chemwiki http://chemwiki.ucdavis.edu/Physical_Chemistry/Spectroscopy/Photoelectron_Spectroscopy/Photoelectron_Spe ctroscopy%3A_Theory 2