Naming Chemical Compounds
advertisement

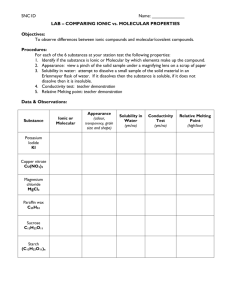
Naming Chemical Compounds “Compound” – pure substance made up of two or more elements chemically combined in fixed proportions. Elements in compounds are held together by chemical bonds which form when atoms gain, lose, or share electrons. If atoms transfer (gain or lose) an electron, then an ionic compound is formed. If the atoms share electrons then a molecular (covalent) compound is formed. Ionic compounds are formed when a metal and non-metal chemically bond. Example: Table Salt (NaCl) When dissolved in water they break apart and release charged atoms called ions. Ex. NaCl + water = solution of ions Na+ (lose an electron) Cl- (gains an electron) The movement of the charged ions in water allows for the movement of an electrical current to flow through the solution. Properties of Ionic Compounds - good electrical conductivity - solid at room temperature - high melting point Naming Ionic Compounds 1. Metal (positive) ion goes first 2. Non-metal (negative) ion goes second and ends in ide Example: NaCl Sodium chloride Ionic Compound **one exception to the second rule is if it is a polyatomic ion (ions with many atoms) ** example: CO3 2- - carbonate CaCO3 = Calcium Carbonate 3. When an element has more than one ionic charge, use Roman numerals to indicate which atom is being used. Example. iron (II) chloride (Fe2+ ion is used in the compound) Molecular compounds are formed when two or more non-metals chemically bond. Example: Sugar (C12H22O11) or Carbon dioxide (CO2) Properties of Molecular Compounds - Poor conductors /good insulators - solid, liquid or gas at room temperature - low melting point When dissolved in water, neutral molecules are released into the solution, which cannot carry an electrical current. C12H22O11 (s) + Water C12H22O11 (aq) Molecular compounds are named by: 1. Writing the name of the first element. 2. Changing the ending on the name of the second element to –ide. 3. When more than one atom is used in the formula, you start each name with a prefix. 1 = mono, 2 = di, 3 = tri, 4 = tetra, 5 = penta example: N2O3 dinitrogen trioxide 4. exception: do not use a prefix if the first element has only 1 atom example: CO2 carbon dioxide Molecular Compound Common Prefixes Common Polyatomic Ions Mono 1 NO3 Nitrate Di Tri Tetra Penta Hexa PO4 Phosphate CO32- Carbonate OH - Hydroxide NH4+ Ammonium ClO3- Chlorate 2 3 4 5 6 3- SO32- Sulphate HCO3- Bicarbonate NAME THE FOLLOWING 1. NaF _________________ 21. HI _________________ 2. NaOH _________________ 22. SrS _________________ 3. KCl _________________ 23. NaCl ________________ 4. FeCl2 _________________________ 24. CO __________________ 5. CrF2 _________________________ 25. HF __________________ 6. TiI4 _________________________ 26. CoCO3 ______________ 7. NH 4OH _______________ 27. Cu(HCO3)2 _________________ 8. PbO _________________ 28. BeO ________________ 9. Mg(NO3)2 _____________________ 29. NH4I ________________ 10. BaO 30. Ca3(PO4)2 __________________ _________________ 11. AgBr _________________ 12. Fe2O3 __________________________ 13. SeF6 __________________________ 14. FePO4 __________________________ 15. SCl4 __________________________ 16. (NH4)2S _________________ 17. MgI2 ___________________________ 18. NF3 __________________________ 19. P4S5 ____________________________ 20. ZnI2 ____________________________