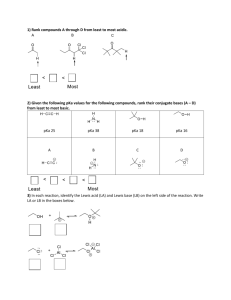
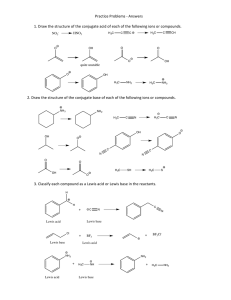
Acid_Base_Sep21 - Blogs@UMass Amherst
advertisement

Acid-Base Chemistry Sept 21 Chapter 6:206 6:206-215 215 HW: 27, 28, 29, 30, 31, 32, 33, 36 Frustrated Lewis Pairs(#36): J. Am. Chem. Soc., 2009, 131 (29), pp 9918–9919 Goals: Predict pKa using periodic trends, structures; solvent levelling What happens when chemical compounds are added to water? 2 ... - 6 6 6UMass 6 6 Amherst Created by Karsten Theis Size and # lp governs acidity in binary hydrides - 6 6 6 6 6 2 ... 2 Question! Which reason explains why HF is a stronger acid than CH4? A) electronegativity of the E-H bond B) delocalizing negative charge in the conjugate base C) the octet rule of the conjugate base D) expanded octet for the binary hydride E) Atomic radius of the central atom - 6 6 6 6 6 2 ... 3 Which statements are correct? 1) 2) 3) 4) pKa rank is HClO4 < HClO3 < HClO2 < HOCl All shown Lewis structures are correct All shown Lewis structures are incorrect Delocalized charge in conjugate base dictates pKa ranking A) 1 B) 1 C) 2 D) 3 E) 1 and and and and and 2 3 4 4 4 - 6 6 6 6 6 2 ... 4 Which statements are correct? 1) 2) 3) 4) pKa rank is HClO4 < HClO3 < HClO2 < HOCl All shown Lewis structures are correct All shown Lewis structures are incorrect Delocalized charge in conjugate base dictates pKa ranking A) 1 and 2 B) 1 and 3 C) 2 and 4 D) 3 and 4 E) 1 and 4 pKa ~ 9 – 7n n = # pi-bonded O-atoms - 6 6 6 6 6 2 ... 5 H+ is a hard acid; so is Fe3+ - 6 6 6 6 6 2 ... 6 Rank the Ksp for these transition metal species in water, and the reason why A) Mg(OH)2 < Fe(OH)3 < Fe(OH)2 < Zn(OH)2 Due to χ of cation B) Fe(OH)3 < Mg(OH)2 < Fe(OH)2 < Zn(OH)2 Due to Q/r of cation C) Zn(OH)2 < Fe(OH)2 < Mg(OH)2 < Fe(OH)3 Due to delocalized charge on lone pairs D) Mg(OH)2 < Fe(OH)3 < Fe(OH)2 < Zn(OH)2 Due to reduction potentials E) Mg(OH)2 < Fe(OH)2 < Zn(OH)2 < Fe(OH)3 Due to covalent bonding - 6 6 6 6 6 2 ... 7 Solvent Leveling Effects: from the conjugate acid or base of the solvent. In H2O, the pKa trend of HX is HI = HBr = HCl < HF ‘strong’ acids form H3O+ (stronger than H3O+) (Is H2O a conjugate acid or base?) In glacial acetic acid (H3CCOOH), the pKa trend is HI < HBr < HCl < HF ‘strong’ strong acids form H3CCOOH2+ (stronger than H3CCOOH2+) - 6 6 6 6 6 2 ... 8