a) SnI Tin (IV) iodide b) lead (II) phosphate Pb3(PO4)2 c) CuClO2
advertisement

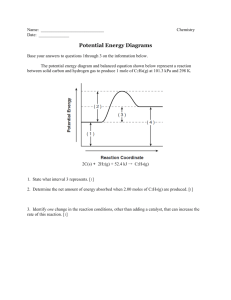
Chemistry - Chap 10- Formative Assessment 1. What is the molar mass of NH4Cl? Name _________________________________ 1N+4H+1Cl=14+4+35.5 = 53.5 g/mol 2. How many molecules are in 3.75 moles of H2O? use Av # so 3.75mol x 6.02 x 1023 molec/mol =2.3 x 1024 3. What is the mass in grams of 2.25 moles of Fe? Use PT s0 2.25 mol x 55.8g/mol = 125.6 g 4. What is the volume of 0.75 moles of oxygen gas (O2) ? use 22.4 so .75 mol sx 22.4 L/mol = 16.8L 5. How many atoms are in 7.50 g of Al? use PT to get to mol then av # so 7.5g Al x 1mol/27gAl x 6.02 x 1023 = 1.7 x 1023 atoms 6. What is the percent composition of H and O in hydrogen peroxide (H2O2)? Part/whole so 2gH/34gcmpd x100=5.9 % so O is 100-5.9 = 94.1 % 7. Give the name or formula for each ionic compound. c) a) SnI4 Tin (IV) iodide b) lead (II) phosphate Pb3(PO4)2 CuClO2 Copper(I) Chlorite d) iron (III) carbonate Fe2(CO3)3 Use the following scale and assess yourself on how well you can completed this activity. Feel free to estimate in between each category. Example: 2.5 if you need help on some, but can do the rest on your own. 1- Below Basic: I am having difficulty in meeting the standard. (I don’t know what I’m doing) 2- Basic: I am approaching proficiency in meeting the standard. ( I could finish with some help) 3- Proficient: I can consistently demonstrate proficiency in meeting the standard. ( I can do this and will be able to do it later) 4- Advanced: I can consistently exceed key concepts, processes & skills. Exceeds the standard ( I can do more questions similar to these) My overall assessment ______________