Energetics and Metabolic Pathways
advertisement

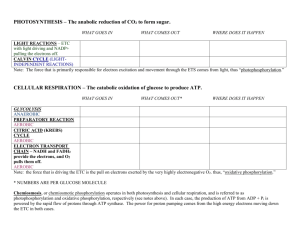
Metabolic Pathways relating to energy: Photosynthesis and Cellular Respiration The following is loaded with the terminology of metabolism and energetics. I have underlined or italicized the key terms. The more you use them (read, write, listen, and speak), the more familiar they will become to you. The concepts are difficult enough without stumbling over the terminology. Read this as often as necessary until you can read all the way through without stumbling. Photosynthesis and Cellular Respiration are very significant metabolic pathways because in these pathways we can see how energy enters the biosphere, how it is stored, how it is transferred from one molecule to another, and how it is used. There are countless other metabolic pathways, but these two are brought into focus because of their significance in energy. Metabolic pathways are called "pathways" because the reactions involve numerous steps, each a chemical reaction catalyzed by a specific enzyme. Each step involves "intermediates" - partially-constructed molecules. Anabolic pathways: When smaller molecules are built up into larger ones the metabolic pathway is anabolic. Photosynthesis is an anabolic pathway, because cells are "building" sugar molecules from carbon dioxide and water. Linking glucose molecules together to make starch is another anabolic process. Anabolic pathways are endergonic. Ever heard of anabolic steroids? What is being 'built up' with the help of anabolic steroids? Catabolic pathways: When large molecules are broken down and their energy is extracted, the pathway is catabolic. In aerobic cellular respiration, the energy contained in the bonds of sugars (and amino acids and fatty acids) is extracted by oxidation… all the way down to carbon dioxide. In digestion, polymers are hydrolyzed into their monomers. Both are examples of catabolism at different levels, and both are exergonic. The pathways of cellular respiration have the ultimate goal of extracting energy from food and transferring it to ATP. It is the same basic process in all life forms from bacteria to humans. Pretty much all of the enzymes involved are the same across all life, and that means that the genes (DNA) coding for those enzymes are also the same. As we will see, there is some wiggle room in the genes coding for the same protein/enzyme, so by analyzing the sequence of A, T, C, and G in the DNA coding for any specific gene, we can infer from accumulated changes (mutations) how distantly related any two organisms are. This topic will have to wait. Oxidation and Reduction: The energetics pathways involve many reactions that only occur together (they are 'coupled'). Oxidation involves the loss of one or more electrons and their energy. An oxidized molecule has less free energy than it had before it was oxidized. The electrons lost by one molecule must be picked up by another molecule. When a molecule receives electrons from another, it is reduced, and its free energy is increased. So whenever a molecule is oxidized, some other molecule is reduced, and vice versa. Because the two processes are inseparable, we often refer to such reactions as REDOX (oxidation/reduction) reactions. You "saw" a redox reaction when you tested reducing sugars with Benedict's reagent. A copper compound is reduced by a sugar molecule (that gets partially oxidized). So in that reaction, a sugar loses an electron (oxidized) and the copper compound in Benedict's reagent gains an electron (is reduced). That's why the test only works for 'reducing' sugars. (Even though the sugar gets oxidized, it was the source of the electrons that reduced the copper compound…. Thus "reducing sugar") There are molecules in both photosynthesis and cellular respiration that are so well-suited to being reduced, oxidized, reduced, oxidized - in an endless cycle, that they are referred to as electron carriers. NADH and FADH2 in cellular respiration, and NADPH in photosynthesis are the reduced forms of the electron carriers. The oxidized forms are NAD+, FAD++, and NADP+, respectively. All 3 are vitamins in the category of B vitamins. In addition to being "electron carriers" and vitamins, these molecules are also referred to as coenzymes, since they work with enzymes to catalyze reactions. Interestingly, all of these electron carriers are like little tiny pieces of DNA or RNA (as is ATP). NADH and NADPH are also chemically related to nicotine (the N stands for nicotinamide). Like the relatedness of organisms that appear to be so different, these related molecules that are found in every cell provide evidence for the common ancestry of all life. Phosphorylation: When a phosphate group (PO4) is bonded to a molecule, the bond is a high-energy bond and the free energy of the recipient is increased. Most commonly, "phosphorylation" refers to the bonding of the third phosphate group to ATP (ADP + P > ATP). The bond between the second and third phosphate groups is the source of most energy used by organisms to do work. Phosphorylation requires energy from food. However, in the light-dependent reactions of photosynthesis, ATP is phosphorylated by the energy from light (photophosphorylation). This ATP is primarily to make sugar, and then the plant can transport the sugar (not the ATP) to its roots, flowers, fruits, and other non-photosynthetic organs. In cyanobacteria (the term applied to all of the blue-green pigmented bacteria of which there are numerous species) the ATP from photosynthesis can be used for cellular work (some ATP) or to make sugar (most). Aerobic or anaerobic respiration: Phosphorylation of ATP in cellular respiration can occur with (aerobic) or without (anaerobic) oxygen. The yield of ATP is much greater in the aerobic pathways of respiration. If we go back to the very origin of life on Earth, we are fairly certain that there was little or no atmospheric or dissolved oxygen. Oxygen is simply too reactive to exist for a significant amount of time. Today, all atmospheric oxygen is constantly being regenerated by photosynthesis. So we can infer that phosphorylation of ATP was primarily anaerobic (w/o oxygen) in the very earliest life forms. On today's earth, we find microbes (bacteria and archaea) that live in oxygen-free environments, and that are in fact killed by oxygen - so both anaerobic and aerobic respiration occur today. At some point - evidence of photosynthetic bacteria dates back 3.8 billion years - the oxygen-producing (photosynthetic) life forms began to proliferate. Oxygen released into the water would react quickly. Anaerobes would be safe in their oxygen-free environments, and the oxygen level was generally very low and localized. Some organisms most likely evolved that could tolerate oxygen. And in fact, with oxygen the efficiency of making ATP from food molecules goes up dramatically. (We know that a single glucose molecule can phosphorylate 2 ATPs without oxygen, and 36 ATPs with oxygen.) For these oxygen-using and oxygen-tolerant microbes, an 18-fold increase in fuel efficiency would have created a significant advantage. Aerobic respiration is such an advantage that all of the multicellular life on the planet today is both oxygen-tolerant, and oxygen-dependent. As for the microbial world, we see today a full spectrum of organisms with regard to their need for oxygen and their ability to tolerate oxygen. Some microbes today are quickly killed by oxygen. These are anaerobic organisms, and as you might imagine, they are found in anoxic (zero oxygen) environments. Oxygen has its drawbacks, even with us. You hear about foods rich in "antioxidants" and that's a good thing. Some vitamins have antioxidant properties. The reason we need antioxidants is that free radicals of oxygen can disrupt DNA and lead to cancer. So even though we must have oxygen to live, it is considered to be the #1 cause of cancer. Let's get back to oxidative (aerobic) respiration. Oxygen exerts a very strong pull on electrons, and it is the electrons moving through the electron transport system of cellular respiration that allows us to phosphorylate enough ATP to keep us alive. The process of producing ATP with oxygen is called oxidative phosphorylation. We can make a little extra ATP without oxygen, and this can help when we are pushing ourselves to the limit. The downside is that lactic acid is a byproduct of anaerobic respiration in muscles, and can build up in muscle tissue resulting in soreness that can last for days. In photosynthesis, electrons are driven off the chlorphyll molecules and pushed down an electron transport system. Eventually, they are pulled off the end of the line by NADPH (see above). The process of producing ATP in the light-dependent reactions of photosynthesis is called photophosphorylation. This ATP, along with the reduced NADPH is used to make sugar in the Calvin Cycle (light independent reactions). Chemiosmosis: There is an electron transport system in both photosynthesis (the light reactions) and aerobic cellular respiration (the electron transport system). They are amazingly similar. Both involve the movement of electrons through molecules embedded in cell membranes (thylakoid membranes in photosynthesis, and mitochondrial membranes in oxidative respiration). In both, the moving electrons provide the energy to pump protons (H+) across a membrane against their concentration gradient, and in both, these protons rush through an enzyme complex called ATP synthase. The rush of protons through ATP synthase provides the energy for phosphorylation of ATP. That's chemiosmosis. "Chemiosmotic phosphorylation" is also called photophosphorylation (in photosynthesis) and oxidative phosphorylation (in aerobic respiration).