Chapter 11, lecture 1
advertisement

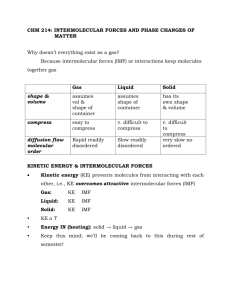
Chapter 11: Intermolecular Forces To this point: we’ve focused on the nature of the forces within molecules E.g. H2O: we can describe the bonding, geometry, and polarity using VB and VSEPR ideas Intramolecular forces forces within molecules, e.g., covalent/ionic bonds, etc. influence molecular shape, bond energies, polarity, etc. Now: how do molecules interact with each other? Intermolecular forces forces between molecules nature of intermolecular forces determines physical properties of liquids & solids, e.g., boiling & melting points What differentiates the gaseous state from the liquid & solid states? 1 How to model the three states of matter? We use the kinetic – molecular theory Kinetic-molecular description of gases, liquids & solids Kinetic-molecular model of a gas: Collection of widely separated, noninteracting particles Constant, random motion Average kinetic energy of particles >> attractive forces between particles Lack of strong attractive forces between particles allows a gas to fill its container 2 Kinetic-molecular model of liquids Intermolecular attractive forces strong enough to hold particles close together Liquids are more dense and less compressible than gases Less empty space between particles than in a gas Clusters or groups of particles can still move past one another Average kinetic energy of particles intermolecular attractive forces 3 Kinetic-molecular model of solids Intermolecular attractive forces strong enough to lock particles in place Less compressible than liquids – less empty space between particles Often have highly ordered structures (crystalline solids) Particles are not free to move - they can only vibrate relative to one another Average kinetic energy of particles << intermolecular attractive forces Because of the close proximity of particles in a solid or liquid (relative to a gas), these phases are referred to as condensed phases of matter 4 How does a substance undergo a phase change (e.g., sl, lg)? How are the attractive intermolecular forces overcome? The physical state of a substance depends on the balance between kinetic energy and intermolecular attractive forces Since kinetic energy depends on T, we can initiate a phase change in a substance by heating/cooling What is the nature of the intermolecular forces that determine the physical properties of liquids/solids? 5 Intermolecular forces Generally much weaker than intramolecular forces (i.e., covalent/ionic bonds) For example: ~ 16 kJ of energy is necessary to overcome the intermolecular attractions in liquid HCl in order to vaporize it (HCl(l) HCl(g)) But… about 431 kJ of energy is required to break the H – Cl polar covalent bond (HCl(g) H(g) +Cl(g)) 6 In liquids: two properties in particular reflect the strength of the intermolecular forces -vapor pressure (later in this chapter) -boiling point In general, the stronger the IMF, the higher the boiling point In solids: the melting point increases as the strength of the IMF increase What types of intermolecular attractions exist between electrically neutral molecules? Dispersion forces Dipole-dipole attractions Hydrogen bonding What kind of attractive force operates between a permanently charged particle (i.e. an ion) and a neutral molecule? Ion-dipole force (important in solutions) 7 Notice: all intermolecular interactions are electrostatic in nature They are therefore governed by a Coulombic force law: So why are IMF so much weaker than ionic (or covalent) bonds? General guidelines… Ionic bond strengths: 700 – 3800 kJ (table 8.2) Covalent bond strengths: 150-1100 kJ (table 8.4) Dispersion force: ~ 2 – 20 kJ Dipole – dipole force: ~ 2 – 20 kJ Hydrogen bonding: ~ 5 – 25 kJ Ion – dipole: ~ 15 kJ Recall: from Coulomb’s law… Attractive force F (between opposite charges): Increases as Q1, Q2 increase Decreases as d increases (a) Charges responsible for IMF are generally MUCH smaller than charges in ionic compounds e.g. NaCl vs HCl 8 and (b) distances between molecules are much larger than distances between atoms (or ions) held together by bonds 1. Dispersion Forces Is there evidence for interactions between neutral nonpolar atoms or molecules? Gaseous helium can be liquified, as can N2(g) , O2(g), etc. At low T, high P (these cases) there must be some type of interaction which overcomes the kinetic energy of the particles and allows them to form liquids These interactions between neutral, nonpolar species are known as dispersion forces (or London dispserion forces) Motion of e- in an atom or molecule gives rise to an instantaneous dipole moment, e.g., 2 He atoms: 9 The 'induced' dipole moment changes over time (as the emove); The distortion of charge distribution sets up the attractive interaction known as the dispersion force (LDF) What determines the degree to which the charge distribution will be distorted? Polarizability: ease with which e- charge cloud is distorted The greater the polarizability, easier e- cloud can be distorted to set up an instantaneous dipole Larger atoms/molecules: larger polarizability (why?) Net result: strength of dispersion forces increases with increasing MW Substance MW (amu) Boiling point (K) F2 38.0 85.1 Cl2 71.0 238.6 Br2 159.8 332.0 I2 253.8 457.6 10 How do the shapes of molecules influence dispersion forces? E.g., which has the higher boiling point: CH3CH2CH2Cl or (CH3)2CHCl? (These species are isomers – same formula, different structure) 2. Dipole - dipole forces Exist between neutral polar molecules Result from electrostatic interaction between positive/negative ends of dipoles; e.g., HCl Dipole - dipole forces effective when molecules are very close together – they are a balance between attractive and repulsive interactions Repulsive interactions force molecules apart – net result is a weak attraction between molecules 11 Note: for molecules of similar mass & size, strengths of attractive interactions increase w/ increasing polarity What effect would this have upon the boiling point of a liquid? Substance MW (amu) Dipole moment (D) Boiling point (K) CH3CH2CH3 44 0.0 231 CH3OCH3 46 1.3 248 CH3Cl 50 1.9 249 CH3CHO 44 2.7 294 CH3CN 41 3.9 355 12 Problems du Jour What is the nature of the major intermolecular attractive forces in Xe(l) PCl3(l) Br2(l) Describe the intermolecular forces that must be overcome to convert each of the following from a liquid to a gas: Br2 H2S CH3CH2CH3 13