Organic Chemistry Notes
advertisement

Summary of Organic Reactions
by F.7B 2000/01
Alkane
General Features
Hydrocarbon
sp3
single covalent bonds = saturated
Each C is tetrahedral
hydrophobic = immiscible with water
general molecular formula = CnH2n+2
Physical Properties of Alkanes
Boiling points
Van der Waals’ forces
molecular size
degree of contact
Chemical Reaction of Alkanes
-
1.Combution of alkanes CH4(g)+2O2 (g)
2.Halogenation
CH4(g)+Cl2 (g) hv
2H2O (g)
CH3—Cl(g)+HCl(g)
Free Radical Mechanism, mixture of products
3.Nitration
4. Oxidation
5. Dehydrogenation
C3H8(g)+HNO3(g) C3H7—NO2(l)+H2O(g)
CH4 + 2 O2 CO2+ H2O
C2H6---850oC --->H2C=CH2+H2 (not in school lab)
MECHANISM OF Free Radical SUBSTITUTION (Halogenation) of ALKANES
Initiation
Cl2 --------> 2 Cl
Propagation
Cl
(chlorine radical)
+ CH4 ------> HCl + CH3
CH3 + Cl--Cl ------> CH3Cl + Cl
Termination
Cl+ Cl ------> Cl2
CH3 + CH3 ------> CH3—CH3
Cl + CH3 ------> CH3Cl
USE OF ALKANES
methane in the form of natural gas is widely used as a fuel but it is also an important sources of
many other chemicals . Ethane is used for making chloroethanes and as a sources of ethene on
dehydrogenation.
Alkenes
YMC&MPY
General Features
1.
2.
3.
4.
Unsaturated
The pair of electrons in theπbond are more diffuse
Cannot rotate about the double bond
If double bond and single bond occurs alternately, resonance will occur.( e.g.
CH2=CH-CH=CH2), stability increases.
Important Points (Exam.)
Geometrical isomers
Rotation of the C-C double bond will destroy theπbond. The restriction to rotation of C-C
double bond give rise to geometrical isomers.
Markownikoff’s Rule
When a non-symmetrical electrophile is added onto the double bond of a non-symmetrical
alkene, the problem of orientation of addition arises.
Markovnikov’s Rule : addition will take place so as to give the most stable intermediate
carbocation.
Addition will take place so as to give the most stable intermediate carbocation.
CH2=CHCH3 + HBr
CCl4
CH3CHBrCH3
Mechanism︰
CH2=CHCH3 + H-Br
CH3CHCH3 + Br
CH3CHBrCH3
Decolourization of permanganate solution
CH2=CH2 + KMnO4/OH
-
cold
Decolourization of bromine water
CH2=CHCH3 + Br2/H2O
CH2OHCH2OH
CH2CHCH3
Br OH
Catalytic hydrogenation
The addition of hydrogen to the C=C double bond always involves catalysis by such metals
as Ni, Pt at room temperature and pressure.
Ozonolysis of alkenes
Ozone, O3, is highly reactive electrophile. When it is bubbled through a solution of an
alkene , it reacts with the C=C double bond to yield a five-membered ring . since
the –O-O-O- is quite unstable, this intermediate immediately rearranges to give the more
stable ozonide. This ozonide then cleaved with zinc in aqueous acid to form aldehydes or
ketones.
Used in the determination of positions of C=C bonds in alkenes
Ozonolysis provides a useful method for determining the position of C=C bonds.
The parent aldene can be reconstructed by recombining the aldehyde or ketone with a double
bond at the carbonyl carbons. For example,
Elimination
alcoholic KOH
(CH3)3C-Br
(CH3)2C=CH2
Polymerization(Free Radical Mechanism)
Alkenes undergo addition polymerization, in which individual monomers are joined together
without the elimination of molecules.
Chain initiation
R•+ CH2=CH2
R-CH2 CH2•
Chain propagation
R-CH2 CH2• + CH2=CH2
R- (CH2 CH2)n-CH2 CH2• (two eqn. needed for exam.)
Chain termination
2 R- (CH2 CH2)n-CH2 CH2•
R- (CH2 CH2)n-CH2 CH2-CH2 CH2- (CH2 CH2)n- R
Use of Polymer
Low density poly(ethane) is made by the ICI high –pressure process. This polymerization
takes place at a high pressure of over 1500atm and a temperature of about 200℃
AROMATIC HYDROCARBONS
General Features
Structure of benzene:
The above is one of the 3 Kekule structures ( Do you know the Dewar Structures )
benzene is a resonance hybrid of all the canonical forms.
Important Points
C6H5-C-(CH3)33 is the only alkylbenzene resistant to side-chain oxidation by hot KMnO4/H+
REACTION
CYCLOHEXAN
E
COMBUSTION
Orange and blue
flame
Very little soot
Appearance of
flame
CYCLOHEXENE
METHYLBENZENE
Orange and blue flame Dark orange flame
Little sootiness
Very sooty
ACTION OF Br2
(in
1,1,1-trichloroethane)
1) in dark
2) in light
3) in dark with iron
powder
Liquid mix
No other change
Liquids mix and
decolourise
Liquid mix
No reaction
Liquid mix and
Liquids mix and
Decolourise
Decolourise
Steamy gas evolved No gas is given off
Which forms white
Fumes with NH3
The gas is HBr
Slight reaction
Some decolourisation of
bromine
No gas detectable
Liquids mix
No other change
Liquids mix
Brown colour fades
rapidly
Steamy gas evolved
Which forms white fumes
with ammonia
Liquids mix and
decolourise
ACTION OF
BROMINE WATER
No reaction
Bromine rapidly
Liquids remain
Decolourised
separate
Liquids remain separate
On shaking,
The brown colour
slowly transfers
from the lower
aqueous to the
upper organic layer
ACTION OF
ACIDIFIED
POTASSIUM
MANGANATE(VII)
Liquids remain
separate
No reaction
No reaction
Liquids remain separate
On shaking,brown colour
transfers from aqueous
layer to benzene
Bromine rapidly
Liquid remain separate
decolourised
No reaction
Liquids remain separate
ACTION OF
Liquids remain
CONCENMTRATED separate
SULPURIC ACID
No reaction`
Liquids mix and react
vigorously
Colour darkens to
charred black mass
Liquid remain separate
No reaction
Liquids remain
separate
No reaction`
Liquids mix and react
vigorously
Colour darkens to
charred black mass
A yellow solution is
formed fairly rapidly
rapidly in the cold
ACTION OF
CONCENTRATED
NITRIC (V) AND
SULPHURIC (VI)
ACID
REACTION OF BENZENE
REACTION
3 CH=CH --------> C6H6
REACTANT USED
nickel cat.
NITRATION OF BENZENE
HNO3 + 2 H2SO4 ------->
NO2+ + H3O+ + 2HSO4S U L P H O NA T I O N
C6H6+ SO3
------> C6H5---SO3- + H+
H A L O G E NA T I O N
C6H6 + 3 Br2 ------> C6H6Br6
F R I E D E L -C R A F T S A L K Y L A T I O N
C6H6 + R+ ------> C6H5---R + H+
F R I E D E L -C R A F T S A C Y L A T I O N
C6H6+ RC+O
Conc.HNO3
ConcH2SO4
50-55 c
ConcH2SO4
80c
reflux (prolonged)
FeBr3
Br2
25oC
Aluminium halide
AlCl3
AlCl3
-----> C6H6--CO-R + H+
O X I DA T I O N O F A L K Y L B E N Z E N E
C6H5-R---------->
C6H5--COOH + carboxylic acids
KmnO4/OHH3O+
Reflux
Halogeno-compounds
General Features
Volatility
For RX compound, when X = Cl or F ,the b.p. will be higher than alkane
which have the same molecular mass due to the dipole-dipole interaction
between the molecules.
Solubility
As the interaction between water and RX are quite different, (H-bond and
dipole-dipole),they are only sightly soluble in water.
Classification The compounds can be classified as primary, secondary and tertiary,
depending on the C atom to which the halogen atom is linked.
Reactions
SN and Elimination .
Chemical Reaction
They undergo two types of reactions:(1) SN reaction and (2) Elimination
Reaction. Both (1) and (2) are brought about by basic reagents, and
hence there is always competition between the two reactions.
(1) SN reaction
e.g. R-L + Nu R-Nu + L(leaving group)
Nucleophiles are characterized by being bases, and leaving groups are characterized
by being weak bases.(i.e. the stronger of two bases is often the more powerful
nucleophile, and the weaker of two bases is often the better leaving group.)
N.B. However, we must notice leaving group and nucleophile are matters of rate while
basicity is a matter of eqlm. To say which Nu is stronger, we compare their attacking
speed to carbon and it is the same for comparing leaving group. And it also depends in
the extent of polarization on α-carbon.
S N1
R-X (slow) R+ + Xr.d.s.
R+ + Nu (fast) R-Nu
Since the reaction is unimolecular , as only one specie is
involved in the r.d.s. ,the overall rate of the reaction is
usually independent to the concentration of Nu but
proportional to the conc. of RX as found from experiment.
An example: (CH3)3CBr + OH- (CH3)3COH
Rate=k[(CH3)3CBr]
S N2
Nu- + R-X [Nu---R---X]- Nu-R + XRate = k[RX][Nu-]
Example:
CH3-Br + OH- [HO---CH3---Br]- HO –CH3 +BrRate = k [CH3-Br][OH-]
Comparing SN1 and SN2
The essential difference between Sn1 and
Sn2 is that Sn1 involves the initial
splitting of the R-X bond, whereas in Sn2 Nu- begin to attach itself to R-X before the
R-X bond is actually splits.
Which one occurs depends on the nature of R, X and Nu- and on the solvent in
which the reaction is carried out. There are also stereochemical issues involved.
1.The nature of R.
CH3-Br
C2H5-Br
(CH3)2CH-Br
------- SN1 rate increase-----
<------ SN2 rate increase------
(CH3)3C-Br
THE RATE OF SN1 IS DETERMINED BY THE STABILITY OF THE INTERMEDIATE,
WHICH IS
Carbocation of the α-carbon. Therefore, 30 RC+ ions undergo SN1 in majority while 10
does not.
The SN2 is inhibited by steric effect as the R group increase in size and by the
increased build up of the –ve charge, through the +I effect of Me groups , on the
α-carbon. This will make the C less open to attack by the OH- in forming
the transition state.
2.The effect of solvent.
If the solvent is itself Nu, it may participate in the reaction as a reagent.
3.The nature of X.
The weaker the C-X bond, the more readily will the sub. take place.
Alternatively, X is a good leaving group if HX is a strong acid, or X- is a weak base.
Therefore, the reactivity increases in passing from RF to RI.Also, it is not easy
to replace –OH, -OR or –NH2 groups as the leaving groups are strong bases. In
acid solution, however, the leaving group are less basic.(H2O, ROH, and NH3)
4.The nature of Nu
Its nature has no effect on Sn1 which are independent of the Nu conc but in
Sn2 the rate does depend on the nucleophilicity of Nu-.
For different atoms within the same periodic table group it is the larger atom
that has the highest Nu strength, e.g.
F- Cl- Br- I---.> increase in nucleophilic power ------.>decrease in basic strength---------
Halobenzene
Halobenzene is unreactive as the C-X bond has multiple bond character due to a
slight extent of overlapping of the p orbital. Also, as sp2 bond is shorter than sp3, C-X
bond is stronger than and hence difficult to react.
Preparation
Reactants
ROH
Products
RCl
Reagents, condition
(1) Bubble with dry HCl
reflux with ZnCl2 as cat.
(2)reflux with PCl5/SOCl2
ROH
RBr
ROH
RI
RCH=CH2
RCH=CH2
RCHBrCH2Br
RBr
RH
RCl
(1) reflux with PBr3
(2) red P + Br2 Distill.
Reflux with I2 +red P.
Distill.
Br2 in CCl4
Pass alkene into HBr in
CCl4
Cl2 in sunlight(gives a
mixture of product)
Reaction of Halogenoalkanes
Reactants
RX
RX
RX
Products
RCN(one more carbon
added)
HX + RNH2
(if not excess, RNH2 further
react with RX to form
R2NH ,then R3N ,an
finallyR4N+X-)
ROH + HX
(if comparing the rate of
hydrolysis add sliver
nitrate(ethanol as solvent),
ppt. formed equ:
HX + AgNO3
For I- --- pale yellow ppt.
For Cl- --- white ppt.
ROH
ROR(ether)
RX(elimination reaction)
RCH=CH2
RX
RX
Reagents, condition
KCN(in alcohol),reflux.
Warm with excess NH3 in
sealed container.
(prevent formation of
mixture of product)
H2O (hydrolysis)
Reflux with NaOH
Na + R’OH ROR’O- + RX ROR
For 10, high tempt.
Reflux with KOH(alc.)
For 30 , dilute OH-.
Summary of halogeno-compound
Characteristics of halogeno-compound:
High polarity of C-Hal due to electronegative difference(EN)
Two types of reactions:
1. Nucleophilic substitution(SN)
Nucleophile: molecule or anion that contains unshared pair of electrons to seek positive
center
[Molecularity is the no. of molecules in transition state in rate determining step]
2. Elimination(E)
Base: attack the positive –carbon
E may compete with SN1
∵Same r.d.s.
CH3X CH2+ + X∴C-X C-Nu (unimolecular nucleophilic substitution)
Alkene (elimination)
1o : SN2
2o : SN2 with E
3o : SN1 with E
E dominant SN -- high temp
--strong base
Haloalkane
Reactions/explanations
Conditions
SN2(Bimolecular)
CH3CH2Br + OHMethyl or 1o
[HO---CH2CH3---Br]CH3CH2OH + BrSN1(Unimolecular)
r.d.s.: CH3CH2Br CH3CH2+ + Bro
Mainly 3
Factors affecting rate of SN
I.
structure of haloalkane
SN1:stability of
carbocation
e.g.alkyl group -electron donating
group intermediate-stabilized by
resonance
relatively unhindered
SN2:steric effect
Nature of nucleophile
Order of nucleophilic ability:
SN1:no effect not involve in RO->HO->>ROH>H2O
r.d.s.
(negative chargeuncharged
SN2:concentration of
molecule)
nucleophile and
nucleophilicity
III. Nature of leaving group
ease of leaving(agree with I->Br->Cl->Forder of bond enthalpy)
uncharged molecule
-better
leaving
group
Protonation(alcohol in acid)
H2O>>OH->ORII.
Rate of reaction:
Unreactivity of Halobenzene
∵C-X bond has multiple bond
character ∴ no SN2
∵Sp2 overlap C-X bond
shorter and bond enthalpy larger
∵If SN1 loss of aromaticity
Elimination
HX + R-OH+ R-X + H2O
(neutral; good leaving group)
SN1+SN2>SN1>SN2
e.g. Ar–CH2X
Ar-COCl (Both O and Cl are
electronegative to withdraw electrons
form C, making C become more
positive)
Halide
Dihalides (-2mole Br)
H-C-C-Br -C=CBr-C-C-Br -C≡C-
Alcoholic NaOH
Excess KOH/C2H5OH
with reflux
Formation of bond
C-C: ↑1 carbon atom
R-C-Br + R-C-CN COOH
C-O bond: alcohol
ether
R-X R-OH
R-X R-O-R
KCN/alcohol;H+
≠HCN
∵weak base→ poor
nucleophile
toxic gas
NaOH/reflux
CH3CH2O-Na+/reflux
(CH3CH2-OH + Na
CH3CH2O- + Na+ + H2
C-N bond
Conc.NH3 mixture of
products may be formed
∵alkyl group bonded to N act as
electron donating group→
nucleophilic power↑
large excess of NH3
Mixture of products
CH3CH2-X + NH3
CH3CH2-NH2 (CH3CH2)2-NH
(CH3CH2)3-N
(CH3CH2)4-N+X-
CN- as base(↑1 C)
NH3 + CH3CH2I CH3CH2-NH2
(monosubstituted) CH3CH2I + CNLiAlH4/ether or 2H2/Ni
CH3CH2CH2-NH2
<<Alcohols>>
LLK
P H Y S I C A L P R O P E R T I E S ~~
1) Melting points, boiling points: Relatively high due to intramolecular H-bonds.
2) Solubility in water: -OH group form H-bond with water molecules, so miscible with water in
all proportions. For members with >6 carbons, degree of significance of -OH group decrease, so
less or not soluble.
3) Highly inflammable for short-chained members.
4) Characteristic deep, broadband between 3300 cm-1 to 3600 cm-1 in IR spectrum due to
presence of -OH group.
5) Relatively neutral.
C H E M I C A L P RO P E RT I E S ~~
Reactions involving breakage of C-O bond:
1) Dehydration of alcohols:
(a) Intramolecular dehydration (alkenes formation):
-alcohol heat with excess conc. sulphuric acid.
-heat with pumice stone ,Al2O3.
ease : 3。>2。>1。
(b) Intermolecular dehydration (ether formation ):
-Excess alcohols heat with conc. H2SO4. (Reversible)
2) Halide formation:
(a) With hydrogen halide:
- R-OH + HX ---> R-X + H2O
-Lucas Reagent ,ZnCl/HCl --(b)
rate of cloudiness exist:3。>2。>1。.
With phosphorus halide:
- 3 R-OH + PX3 ---> 3 R-X + H3PO3
- R-OH + PX5 ---> R-X + POX3 + HX
Reactions involving breakage of O-H bond:
1) Reaction with active alkali metals (alkoxide formation):
- R->-OH <----> R->-O- + H+
- the alkoxide ion formed is destablised by the +ve inductive conferred by the electron
donating alkyl group, equilibrium position shift to the left.
2) Esterification:
- Formed by refluxing with carboxylic acids, acyl chlorides, acid anhydrides under catalyst of
conc. H2SO4.
3) Oxidation:
- oxidizing agents commonly used:
KMnO4/H+ (aq), (violet to colorless)
K2Cr2O7/H+ (aq). (orange to green)
- 1。alcohol ---> aldehyde ---> carboxylic acid
- 2。alcohol ---> ketone
- 3。alcohol ---> no reaction.
- Green colour changes from orange forms the basis of
the breathalyser.
4) Triiodomethane formation (Iodoform test):
- alcohols with -C(CH3)H-OH group + I2/NaOH (aq)
---> R-COO-Na+ + CHI3.
<<Phenols>>
P H Y S I C A L P RO P E RT I E S ~~
Solubility in water: slightly dissolved.
Relatively acidic.
The lone pair of O is partially overlap with the ring system, this multiple bond character confers
it a strong C-O bond.
C H E M I C A L P RO P E RT I E S ~~
1) Alkoxide formation:
- The negative charge is dispersed throughout the phenoxide ion(conjugate base) by
conjugation with the π- e- of the ring, so it's well stabilized and more acidic.
- Due to the acidity, it react with NaOH(aq) but alcohols do not, reaction with NaOH(aq)
can be used to distinguish between them.
2) Other reactions of the -OH group:
- Halide formation: slow/no reaction.
- Oxidation: complex polymers formed.
- Elimination: product is not stable due to high angle strain.
- Esterification: slow reaction. Can be modified by using acyl chlorides instead of acids and
converting phenol into phenoxide ion.
- Reaction with NaHCO3: no reaction but carboxylic acids do, a means for distinguishing
the two.
- Reaction with neutral Iron(Ⅲ) chloride: Violet coloration. (Identification of phenols.)
Topic: Hydroxy Compounds
1.1 General properties of Hydrox Compounds
LSC
Alcohol: The hydroxyl group(OH) is attached to a sp3 C
atom
CH3
CH3
C
CH3
tertiary(3。) ROH
3C attached to the C containing OH
OH
2-methylpropan-2-ol
CH3CH2 CH
CH3
secondary(2。)ROH 2C attached to the C containingOH
OH
butan-2-ol
CH3CH2CH2 OH primary (1。) ROH 1 C attached to the C containing OH
propan-1-ol
Phenol: OH is directly attached to a sp2 C atom in an aromatic ring.
OH
OH
phenol
OH
benzene-1,4-diol
In alcohols R-O-H, the O is sp3 hybridized (similar to H2O)
In phenols Ar-O-H, the O is mainly sp2 hybridized because the lone pair of O has to overlap
with the p orbitals of the benzene ring so that the C-O bond has some double bond character.
Therefore, It is more difficult to break.
Due to the presence of the polar O-H group & the ability to from hydrogen bond with water,
alcohols with 1 to 6 C atoms are very soluble in water. Phenols are slightly soluble in water.
Hydrogen bonding:
alcohol & phenol molecules can form intermolecular H-bonds. Then, It has usually higher
melting point & boiling point than other carbon compounds with the same no. of carbon
atoms(except RCOOH).
1.2Preparation of ROH:
A. Hydrolysis of RX
KOH(aq), reflux
(1。/ 2。)R-X(l) + OH (aq)------------------------> (1。/2。)ROH (aq) + X (aq)
AgNO3(aq)/ethanol, warming
(3。)
R-X(l) + H2O(aq)---------------------------------> (3。)ROH(aq) + HX(aq)
{3。RX undergo elimination easily if heating under reflux with KOH(aq) in
addition to substitution . It is better to warm with AgNO3(aq) + ethanol mixture.}
B. reduction of ketone , aldehyde & carboxylic compound (NOT for 3。
ROH)
O
LiAIH4/dry ether ,H2O
OH
ketone R-C-R`------------------------------------- R-CH-R`
2。
aldehyde, RCOOH, RCOOR`----------------------- 1。ROH
LiAIH4 is a very powerful reducing agent.
> heating is usually not required because the reaction is already vigorous(especially for aldehydes)
O
LiAIH4 can also reduce RCOOH,RCOOR`,R-C-CI
LiAIH4 does not react with C=C bonds
NaBH4 only react reduces aldehyde & ketone
C. Hydration of alkene:
H2SO/cold
OSO3H
H2O
OH
R-C=C-R`
-----------
H H
R- C-CH2R
--------- R-C-CH2R`
H
H
D. From 1。 aliphatic amine:
RCH2NH2 +NaNO2/HCI-- RCH2OH +N2(effervescence)
1.3 Test for Phenol & ROH:
ROH & Phenols react with Na but NOT Mg
(RCOOH react vigorously with both Na and Mg)
ROH do not decolorize cold Br2/CCI4 or Br2(aq) in the absence of light but
>phenols
decolorize Br2(aq) with the formation of a white ppt. at room temperature (Note that
phenylamine also react with Br2(aq) to give a white ppt.)
>Phenol can form a coloured( usually violet )complex with neutral FeCI3(aq).
Water soluble ROH (1 to 6C) can be distinguished by the Lucas test(by mixture with saturated
ZnCl3 + conc. HCI)
~~3。 turn cloudy at once.
~~2。 turn cloudy at about 5 minutes (not precipitation)
~~1。 does not change( only dissolves in the acid, due to the formation of ROH2 positive ion).
> ROH (RCOOH ,NOT phenols) react immediately with PCI5 to give HCI(g) which fumes
with NH3(g)
CH3
O
>-CH-OH group(& -C-CH3 too ) can be identified by the iodoform test(I2/NaOH) BECAUSE
OF formation of yellow ppt.of CHI3.
>Further reaction for ROH & phenol:
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
~~~~~~~
Method
|
ROH
|
phenol
|
+acidified K2Cr2O7
| no observable change
| acidified K2Cr2O7
from orange to green
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
~~~~~~~
+NaOH+
|
|
|
BENZENEDIZONIUM
| orange ppt
| no observable change |
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
~~~~~~~
1.4 Preparation of Phenols
FROM diazonium salt ( an important intermediate in synthesis )
The diazonium ion will begin to breakdown with the resulting formation of phenol and
effervescence of nitrogen at above 278K.
N2
-------
OH
dil H positive
above 5.`C
1.5 reaction phenol
phenol reacts with NaOH .Therefore, phenol is a much stronger than alcohol.
OH
O Na +
+ Na OH ------
+H2O
alcohols do not react with NaOH, 1to 5 Carbon alkanols are only miscible NaOH(aq).
The fact that phenol is soluble in Naoh(aq) can be used in isolation of phenol from other organic
compounds(except RCOOH).
1.6 Acidity of Alcohols & Phenols
AC I D I T Y:
OH
BA S I C S T R E N G T H :
H2O is more acidic than ROH because RO
> H-O-H > R-OH
O
< H-O
<R-O
is less stable than OH .
Phenol is more acidic than ROH because a phenoxide ion is more stable than an alkoxide ion
due to the delocalization of e while the e donating alkyl group in ROH destabilizes the
alkoxide ion.
1.6 ESTERIFICATION
alcohol react with mineral acid or organic acid (with H2SO4(l) or dry HCIas catalyst ) to give
ester& water
Ester can be found when alcohol reacts with acid chloride or acid anhydride.
[no catalyst].
~ LEE SUN CHUN 7B 23~
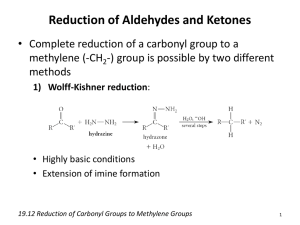
SUMMARY OF REACTIONS OF ALDEHYDES AND KETONES
R E AC T I O N S /R E AG E N T S A L D E H Y D E S
KETONES
Nucleophilic Additions
HCN
NaHSO3
methyl
addition occurs
addition occurs
addition occurs with
Ketones only, not for
other
Specially hindered ones.
Condensation Reactions
Substituted ammonia
Derivatives, e.g.
2,4-DNP
condensation reactions occur
Oxidation
KMO4/H⊕
oxidize, gives
gives acids with same C no.
more
difficult
to
acid with fewer C no.
(C-C bond
breakage)
Tollen’s reagent
Fehling’ solution
silver mirror formed
red ppts. with aliphatic
Aldehydes
no reaction
no reacion
reduced to 10 alcohols
reduced to 20 alcohols
occurs only with CH3CHO
occur
Reduction
LiAlH4 or NaBH4
Special Reactions
Triiodomethane
having
(I2/OH)
with
ketones
CH3•CO group
Observations
Test
ETHANANL
PROPANONE
Addition Reaction with
hot.
Sodium Hydrogensulphate (IV)
precipitate
2 cm3 of carbonyl compound added
dropwise
shakes the tube and cool under a
stream of cold water
2 cm3 saturated NaHSO3 solution
Condensation Reaction with
yellow
Tube became hot.
Tube became
Some milkiness which
White
soon disppreared.
(With methanal and
ethenal the addition
products are so soluble
that they
rarely crystallize)
Formed.
Orange yellow
Orange
2,4-Dinitrophenylhydrazine
precipitate
precipitate
Orange solution
turned blue/green
No change
2. With Fehling’s solution
Add Fehling’s solution into Carbonyl
compound dropwise until ppt. dissolve
and warm for 5-10 mins till no further colour
change
Blue Solution
formed a reddish brown
ppt
No change
3. With Tollens’ reagent
NH3 solution added drop by drop until the
Ag2O ppt. is nearly dissolved and add
1 cm3 of 0.05M AgNO3 with 3/4 drops
of NaOH after mixing with the carbonyl
compound, warm it
Grey precipitate
Silver is usually
produced too rapidly
to form a good silver mirror
No change
2 cm3 of 2,4-Dinitrophenylhydrazine
+ 1-2 drops of the liquid carbonyl compound
Oxidation Reactions:
1. With Acidified Dichromate (VI)
5 drops of carbonyl compound
+ 2 drops of K2Cr2O7
+ 10 drops of dil. H2SO4 (warm
with hot water)
TRIIODOMETHANE REACTION PALE YELLOW PRECIPITATE
PPT
5 drops of carbonyl compound shaken with
1 cm3 I2 solution and NaOH added drop by
drop until colour of I, just disappears
Formed
PALE YELLOW
formed
S.H.H. LEUNG KWAI YEE SECONDARY SCHOOL
AL ORGANIC CHEMISTRY
REVISION NOTES
Aldehydes and Ketones 請用整頁模式
Aldehydes have the general formula ROCH and ketones ROCR'. Carbonyl
group has a noticeable effect on the properties of these compounds. Both
compounds have a planar structure because the carbonyl carbon is sp 2
hybridised.
Trigonal planar structure of ketones or aldehydes
Since oxygen is more electronegative than carbon, the carbonyl carbon is
electron deficient and the carbonyl oxygen is electron rich. Furthermore,
the carbonyl group, being flat, is open to unhindered attack from above
and below in a direction perpendicular to this carbonyl group plane. The
carbonyl group should, therefore, be highly reactive. The reactions of
carbonyl group generally involve the formation of bonds with the
election deficient carbonyl carbon. The reagents, which attack the
carbonyl group will, therefore, be electron rich (nucleophile). In other
words, reduction of aldehyde and ketone will be initiated by nuclophile,
and most of the reactions are nucleophilic addition. The general pattern
of nucleophilic addition may be represented as follows:
N.B:
X- is a nucleophile
All the above processes are reversible. It should be
noted that
(a) It is a nucleophilic addition and followed by protonation.
(b) There is no net reaction.
(c) It is a nucleophilic addition and followed by an elimination.
Usually aldehydes and ketones most commonly
react by the mechanism (a).
The carbonyl group of ketones and aldehydes shows a strong IR absorption
at around 1700 cm-I. This is due to a C = 0 stretching vibration. The IR
spectrum of propanone is shown as follows
R E AC T I V I T Y O F C A R B O N Y L C O M P O U N D S
The reactivity of carbonyl compounds are affected
by
(a) Electronic effect
(i)
Electron-withdrawing group makes the carbonyl carbon
more electron deficient and this facilitates the attack by
nucleophiles.
(ii) Electron-releasing group decreases the electron
deficiency of the carbonyl carbon atom.
Consequently carboxylic acids and their derivatives such as esters
and amides are less reactive than ketones and aldehydes towards
nucleophilic attack because they contain electron-releasing groups.
On the other hand, acid chlorides are very reactive towards nucleophiles,
because chlorine is very electronegative and makes the carbonyl
carbon more deficient.
(b) Steric effect
If the substituents bonded to carbonyl group are very bulky, it will hinder
the approach of the nucleophiles. Consequently, aldehydes are more
reactive than the corresponding ketones.
In short, the reactivity of carbonyl compounds towards nucleophiles is
outlined as follows
R-COCI >
R-CO-H
>
>
RCOR
RCONHR
>
RCOOR
Preparation of aldehydes and ketones
The carbonyl group is an extremely important functional group, and indeed
the chemistry of carbonyl compounds is virtually the backbone of
synthetic organic chemistry. As the aldehydes and ketones can be readily
oxidised to carboxylic acids with common oxidizing agents, so they can
be reduced to alcohols by metal hydrides like NaBLI4 and LiAlH4. The
preparation of aldehydes and ketones are as follows:
L Aldehydes
(a) Oxidation of primary alcohols
Aldehydes are obtained by the oxidation of primary alcohols,
provided that the aldehydes, as soon as it is formed, is removed
from the reaction mixture by distillation, and use of excess
oxidising agent is avoided, otherwise the aldehydes will be
oxidised to carboxylic acids. The common oxidising agents
are acidified dichromate and acidified permanganate. For
example,
CH3CH2OH + K2Cr2O7 / CH3COOH CH3CHO(heating is
required)
(b) Catalytic dehydrogenation of primary alcohols
Aldehydes can be obtained by passing vapour of primary
alcohol over finely divided copper at 300-5000C. For example,
CH3CH2OH+Cu at 3000c CH3CHO
(c) Oxidative cleavage of alkenes
This reaction has been discussed in details in Chapter 4 (See
pg~ 54). A reductive condition should be employed for
synthesis of aldehyde. For example,
CH3
CH3
C =CHCH3 +O3+ Zn +H2O
CH3CH2
C =O + CH3CHO
CH3CH3
(d) Reduction of acid chlorides
Acid chlorides can be reduced to aldehydes when they are treated
with hydrogen in the presence of palladised barium sulphate
catalyst (Rosenmund reaction). The activity of the catalyst is
usually moderated by the poison - sulphur and the reduction does
not proceed to the alcohol stage. For example,
\
CH3COOH+SOCl2CH3COCl+H2Pb/BaSO4.SCH3COH+HCl
Reduction of acid chloride to aldehyde can also be brought out
by a specific reagent, tri-tert-butoxy lithium aluminium hydride, LA
lH(0-t~449)3.
IL
Ketones
(a) Oxidation of secondary alcohols
The reaction is similar to the oxidation of primary alcohols.
Common oxidising agents may be used.
(b) Oxidative cleavage of alkenes
R1
R3
C=C
R2
+
O3
+
H2O2
R1COR2 +R3COR4
R4
(c) Hydration of alkynes
Mercury salt is used as catalyst. For example,
CH3C CH Hg2+,dil. H2S04 --
CH3COCH3
(d) Reaction between organocadmium compounds and acid
chlorides
Ketones can be prepared from organocadmium compounds with acid
chlorides:
(CH3)2Cd + 2C2H5COCl ~ 2C2H5COCH3 + CdCl2
Organocadmium compounds are usually prepared from the
corresponding organomagnesium compounds (Grignard
reagents) by reaction with dry CdCI2:
2RMg X + CdCl2 R2Cd + 2MgXCl
Organocadmium compounds are less reactive than RMgX and
they do not further react with ketone to form alcohol.
Furthermore, organocadmium compounds unlike
organomagnesium compounds, they do not react with functional
groups like
-CN, -COOR, -N02 etc. For example
R2Cd + 2R'CCI 2R'OCR + CdCI2
It should be pointed out that only organocadmium compounds
containing primary alkyl groups are stable. The above reaction
is successful with such organocadmium compounds only.
Reactions of aldehydes and ketones
According to the revised syllabus , the following reactions are
included
CARBONYL COMPOUNDS
N E C L E RO P H I L I C A D D I T I O N
A D D I T I O N E L I M I N AT I O N
MECHANISM OF ADDITION OF
HCN
L
Nucleophilic addition
R E D OX
Aldehydes are usually more reactive than ketones largely because of
the inductive effect and steric effect. Another point should be
mentioned is the reaction medi~ Nucleophilic addition of carbonyl
compound can occur in either acidic or alkaline medium. An alkaline
medium increases the concentration and reactivity of the nucleophile.
For instance, cyanide ion is more reactive than its conjugate acid,
hydrocyanoic acid, in alkaline medium; hydrocyanoic acid is
neutralized and exists in the form of anion.
An acidic medium is capable of increasing the electron deficiency of
carbonyl carbon because
H+ +
C=O
C=O+H
The protonated carbonyl group should be more electrophilic than the
neutral carbonyl group. Therefore, it is easier for the nucleophile to
attack the carbonyl carbon. On the other hand, an acidic medium
decreases the concentration of nucleophiles. which are derived from the
dissociation of weak acids. (e.g.CN- from HCN)
The common nucleophilic addition for carbonyl compounds may be
classified as follows:
OOH
RCOR’ +
CN-
RCR’ + H+
CN
(a) Formation of cyanohydrin
RCR’
CN
Cyanide ion can be added to the carbonyl group of aldehydes and
ketones to yield compounds known as cyanohydrins:
OH
CH3COOH
OH
CH3CHCN
CH3CHCH2NH2
(Cyanohydrin)
OH
CH2=CHCN
On hydrolysis, cyanohydrin yields ot-hydroxyacids cyanohydrin and is
thus a very useful intermediate compound in organic synthesis. For
example,
(b) Bisuiphite addition
Sodium bisulphite adds to most aldehydes and unhindered ketones
(especially methyl ketones) to form bisulphite addition products,
adduct. The formation of the adduct is highly sensative to steric
hinderence.
On cooling the sodium salt of the bisulphite addition compound is
crystallized. This reaction is a method for purification and separation of
suitable carbonyl compounds (ketones and aldehydes) from non-carbonyl
compounds. As the addition is reversible, the addition of acids or bases
destroys the bisulphite ion in equilibrium with the addition compound,
and carbonyl compound is regenerated.
(c)
Addition of alkanols
Alcohols are added to the carbonyl groups in the presence of acid to form
acetals or ketals, which are only stable in alkaline solution. Carbonyl
compounds are regenerated on acid hydrolysis.
RCH=O +R’OH
OR’
with dry ammonia and dil. H+
R
OH
C
+H+
R
C
H
OR’
H
OR’
(hemiactal)
(acetal)
(d) Addition of ammonia and its derivatives
A wide variety of substance with -NH2 group reacts with aldehydes and
ketones by an addition-elinanation sequence to give ~C = N
compounds and water. This reaction usually requires acid catalyst. The
derivatives of ammonia that can undergo this reaction are listed in the
following table.
Table 9.1 Derivatives of ammonia
NH3
NH2OH
H2NNHC6H5
H2NNH-
H2NNHCNH2
NO2
NO2
Ammonia
hydroxylamine
phenythydrazine
2,4-DNPH
semicarbazine
The mechanism is similar to the acetal or ketal formation:
The derivatives prepared from 2,4 - dinitrophenylhydrazine and carbonyl
compounds are usual crystalline compounds with well defined melting
point. The derivatives are, therefore, very useful for identification and
characterization of aldehydes and ketones. It should he noted that there is
no addition-elimination between carbonyl compound and the nitrogen
containing compounds in the above table except ketones and aldehydes.
Furthermore, as these derivatives can be decomposed by boiling with
dilute acids to regenerate the parent carbonyl compounds, they are also
used for the purification of carbonyl compounds.
The condensation products between carbonyl compounds and other
nitrogen containing compounds in the above table are similar except
ammonia. The mechanism of the reaction is similar as shown below:
The aldehyde-ammonia adducts usually are not very stable. They readily
undergo polymerization and dehydration. For example,
IlI
Reduction
(a) Hydrogenation
The carbonyl group is reduced to hydroxyl group by catalytic
hydrogenation. More vigourous reaction condition is required
for the hydrogenation of carbonyl group than a double bond:
(b) LiAIH4 and NaBH4 reduction
Lithium aluminum hydride is a very powerful reducing agent,
which can reduce ketones, aldehydes, esters and acids to
alcohols Rowever, sodium boron hydride is a mild reducing
agent, which can reduce ketones and aldehydes only, but it has
no effect on other carbonyl compounds. Both Lyk1R 4 and
NaBR4 have no effect on the carbon-carbon double bond.
(c) Clemmmensen reduction
Ketones or aldehydes are reduced to hydrocarbon by
amalgamated zinc in the presence of concentrated hydrochloric
acid.
Only ketones and aldehydes are reduced but not other
carbonyl compounds.
Distinguishing between aldehydes and ketone
Most of the reactions described above take place
with both aldehydes and ketones.
There are a number of tests, however,
to distinguish between these classes
of compounds. The basic difference
between aldehydes and ketones is
that aldehydes show reducing
properties but ketones do not. The
common simple tests used for
carbonyl compounds are stated
below.
(a) Brady's reagent
2,4~dinitrophenylhydrazine dissolved in either alcohol or dilute
hydrochloric acid may react with both ketones and aldehydes to form
an insoluble yellow or red precipitate. The structure of carbonyl
compound may be deduced by testing the melting point of the
precipitate after recrystallization. Usually mixed melting point
methed is used for an accurate result. For example,
(b) Tollen's reagent (Silver mirror test)
Tollen's reagent is freshly prepared silver nitrate in alkaline ammonia
solution. It is a mild oxidising agent which oxidises aldehydes but
leaves most ketones untouched. If the reagent and aldehyde are
carefully mixed in a test tube the metallic silver will deposit on the
walls to form a mirror. It should be noted that the test tube must be
clean.
RCOH+2Ag(NH3)2 + + 2OH RCONH4 + H20 + 3NH3 +Ag
Both aliphatic and aromatic aldehyde are able to show positive silver
mirror test
(c) Fehilng's reagent
It is similar to Tollen's reagent. It involves the reduction of
complexed copper(U) ion to copper(I) oxide, which is a red
precipitate. it is also used to test the presence of ftee aldehyde group
in a sugar molecule.
RCOR + 2Cu2+ + NaOH + H20 RCO2Na + 4H+(aq) + Cu20
Ketones and aromatic aldehydes cannot reduce Fehling's solution.
AW
Carbolic Acid and their Derivatives
Formation of Carboxylic acids:
(1) Hydrolysis of Nitriles
RCN RCOOH + NH4+
RCN RCOO- + NH3
(2) Oxidation of alcohols and aldehydes
RCH2OH RCOOH
RCHO RCOOH
(3) Oxidation of Alkylbenzenes
Ar-R Ar-COOH
(1) H3O+ ,reflux
(2) acid hydrolysis
(1) OH- , reflux
(2 ) alkali hydrolysis
KmnO4/H+(aq)
or K2Cr2O7/H+(aq)
(1) KMNO4/H+
(2)heat
(4) Oxidation of Methyl Ketones and some Alcohols
OH
O
|
||
--C—CH3 + 3I2 + 3OH- --C—CI3 + 3I- + 3H2O
O
||
--C—O- + CHI3
The resulting carboxylate has one carbon less than
the original carbon skeleton
(1) OH-
R E AC T I O N O F C A R B OX Y L I C AC I D S
<1>Acidity of carboxylic acids
(1)The several factors affect the acidity of the organic compound:
the strength of the H-A bond
the electronegrativity of A
factor stabilizing its conjugate anion A- with respect to HA
(2)The order of acidity:
RCOOH > Ar-OH > R-OH
(3)E-group can increase acid strength, as the OH bond is weakened
and the acid anion is stabilized.
(5) The inductive effect on acidity will decrease rapidly when the
substituents are placed father away from the carbonyl group.
<2>Reduction with Lithium Tetrahydridoaluminate
R-COOH RCH2OH
(1)LiAlH4/anhydrous ether
Primary alcohol
(2)H3O+
The unsturated acids are unaffected by it
<3>Conversion to Acid Derivatives
(1) Conversion to acyl chlorides
PCl5
Or
RCOOH + SOCl2
RCOCl
Or
PCl3
(2) Conversion to acid anhydrides
RCOOH + R’COCl RCOOCOR’ + HCl
(1)pyridine
(3) Conversion to amides
RCOOH + NH3 RCOO-NH4+
RCONH2 + H2O
(4) Conversion to esters
RCOOH + R’OH RCOOR’ + H2O
(1)excess RCOOH
(2)reflex
(1)H3O+
(2)reflex
The yield of the ester can be enhanced by :
(a) increasing the amount of carboxylic acid or alcohol
(b) increasing the amount of inorganic acid
Some specimen results of the investigation of the reactions of ethanoic acid
A. pH of aqueous solution
Orange-red-pH3-4
B. Reaction with sodium
Hydrogencarbonate solution
C.Reactionwith sodium
D.Reaction with phosphorus
pentachloride
E.Reaction with
2,4-dinitrophenylhydrazine
Gas evolved which turns limewater milky-CO2
Gas evolved which popped with a lighted splint-H2
Steamy gas evolved which gave white fumes with
ammonia-HCl
No change
No change
F.Triodomethane reaction
Reaction of Acyl chlorides:
(1) Hydrolysis with water
CH3-COCl + HCl CH3COOH + HCl
(2) Ester formation with alcohols
CH3COCl + CH3OH CH3COOCH3 + HCl
CH3COCl + Ar-OH CH3COOAr + HCl
Ar-COCl + Ar-OH Ar-COOAr +HCl
(3) Amide formation with ammonia and amines
CH3COCl + 2NH3 CH3CONH2 + NH4Cl
Ar-NH2 + Ar-COCl Ar-NHCO-Ar + NaCl + H2O
+NaOH
(4) Anhydride formation
RCOCl + R’COO-Na+ RCOOCOR’ + NaCl
R E AC T I O N O F AC I D A N H Y D R I D E S
(1)Hyduolysis with water
(RCO)2O + R’OH RCOOR’ + RCOOH
(2)Ester formation with alcohol
(RCO)2O + R’OH RCOOR’ + RCOOH
alkaline
medium
(3)Amide formation with ammonia and amines
(RCO)2O + 2NH3 RCONH2 + RCOO-NH4+
(RCO)2O + 2R’NH2 RCONHR’ + RCOO-R’NH3+
REACTION OF ESTERS
(1) Hydrolysis of ester
RCOOR’ + H2O RCOOH + R’OH
RCOOR’ + H2O RCOO- + R’OH
H3O+ /reflux
OH-/reflux
(2) Reduction to alcohols with lithium titrahydridoaluminate
RCOOR’ RCH2-OH + R’OH
(1)LiAlH4/dry ether
(2) H3O+
R E AC T I O N O F A M I D E S
(1)Hydrolysis
RCONH2 + OH- RCOO- + NH3
(reflux)
+
+
RCONH2 + H3O RCOOH + NH4
(reflux)
(2)Dehydration – nitrile formation
Ar-CONH2 Ar-CN + H2O
-H2O
(1)P2O5
(2)reflux
(3) Hofmann degradation
CH3CH2CONH2 + 4KOH + Br2 CH3CH2NH2 + K2CO3 + 2KBr + 2H2O
One carbon atom less
(4) Reduction to amines with lithium tetrahydridoaluminate
RCONH2 RCH2NH2
(1)LiAlH4
(2)dry ether