2-3b Aldehyde and ketone Overview
advertisement

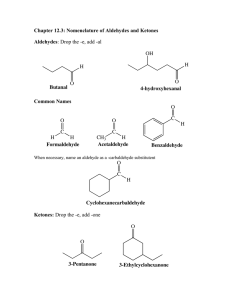
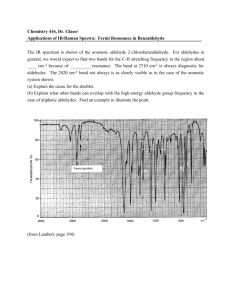
Aldehydes and Ketones Page Reference: 49-56#1-9 Aldehydes and Ketones: Functional Group: Name: Structure: Difference between Aldehydes and Ketones: Rules for Naming: • Find longest continuous chain of carbons to which the =O is bonded. • If it is on an end carbon it is an aldehyde. Name it as an alkane, changing the ending to –al. Groups are added as in alkanes. • If it is on an interior carbon, it is a ketone. Number the chain so that the =O is on the lowest possible number. • Name the compound changing the ending to –one. Location of =O is indicated with a number before the name. Groups are added as in alkanes. Examples: CH3-CH2-CH2=O propanal (No number is needed since it must be a 1) -------------------------------------------------------------------------------------------------------------------CH3 or -Contains a = O on an inner C so it is an ketone -Longest chain: 4 carbons butanone | Number to get OH on lowest (O on 2nd C) 2-butanone CH3 -CH-C=O Side groups: CH3 on 3rd C 3-methyl-2-butanone | CH3 Or --Contains a =O on an end C so it is an aldehyde -Longest chain: 5 C pentanal -side group: ethyl on 2nd C 2-ethylpentanal Questions: 1. In the last example above, explain why we did not call it an hexanal, even though there is a 6 carbon chain. 2. How can you tell the difference between an aldehyde and a ketone. 3. Describe the physical properties of aldehydes and ketones as compared to their parent alkanes and alcohols: boiling point, solubility in water. Explain these physical properties. 4. Describe the following reactions involving aldehydes and ketones and write an example of each: a. Preparation from alcohols b. Conversions to alcohols c. Combustion 5. Explain why primary secondary and tertiary alcohols produce different substances from an oxidation reaction.