Name
advertisement

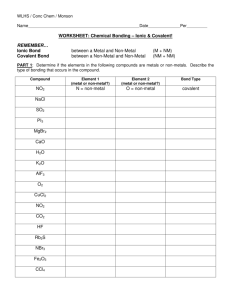
Name: _________________________________________ Date:____________________________ Homework 6: 1.) A 50.0 mL sample of very dilute HCl and 50.0 mL of very dilute NaOH are at 19.50oC to begin with. After mixing the two in a styrofoam cup, the temperature rises to 21.21oC. Calculate the heat of the reaction. (HINT: the density of the HCl, NaOH, and the mixed solutions can be assumed to be that of water 1.00 g/mL) C = 4.184 J/goC qrxn = - qH2O 50.0 mL of water from HCl + 50.0 mL of water from NaOH = essentially 100.0 mL of water! 100.0 mL x 1.00 grams/mL = 100. grams of water qwater = mwaterCwaterTwater qwater = 100. grams (4.184 J/goC)(21.21oC – 19.50oC) qwater = 100. grams (4.184 J/goC)(1.71oC) qwater = 715 J Since the qwater is equal but opposite in sign of the qrxn since the qwater = 715 J then the qrxn = - 715 J 2.) Using the electron configuration for Zn, draw all the orbital box diagrams. Write the complete set of quantum numbers for electrons 1, 5, 9, 21, and 26 1 5 1s 9 2s 2p 3s 21 3p n, l, ml, ms 1st: 1, 0, 0, + ½ 5th: 2, 1, -1, + ½ 9th: 2, 1, 0, - ½ 21st: 3, 2, -2, + ½ 26th: 3, 2, -2, - ½ 4s 26 3d 3.) Define Electronegativity: the propensity of an atom to draw the electron density towards itself in a compound. 4.) Define Electron Affinity: when an atom is able to completely take away an electron from another species and become a negatively charged ion (textbook definition: the energy change associated with one mole of electrons being added to one mole of gaseous atoms or ions) 5.) Which species is the most X atom on the periodic table? Which species is the least? – Given that – what is the trend for X values? F ____ ______ most X ____Fr and Cs_______ least X Circle the correct answer: X values increase X values increase decrease going down a column decrease going from left to right across a row (main group!) 6.) Given the following compounds, classify them based on their bonding as containing ionic, polar covalent, or non-polar covalent bonds: a. GaAs : gallium is a metal and arsenic is a non-metal, therefore predict IONIC b. NH3 (NH bonds only): both are non-metals, not same non-metal and not CH, therefore POLAR COVALENT c. BaO: metal + non-metal therefore IONIC d. SO2 (SO bonds only): non-metal (S) + non-metal (O) not same non-metals and not CH therefore POLAR COVALENT e. ZnO: metal + non-metal therefore IONIC f. NaH: metal + non-metal therefore IONIC g. O2: non-metal + non-metal and same bonded to itself therefore NON-POLAR COVALENT 7.) Work on your Lewis structures handout!! Pages 1-9!