Ionic or Covalent
advertisement

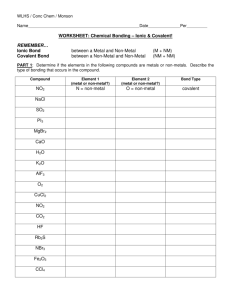
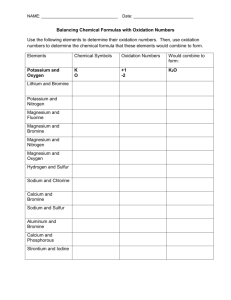
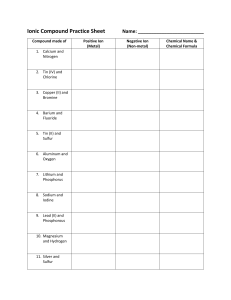
IONIC OR COVALENT? (Page 56) This one will be quick. Just look at the two elements in the compound and tell me if it's an ionic or a covalent compound. 1. 2. 3. 4. 5. Mercury and Oxygen Carbon and Oxygen Carbon and Iodine Beryllium and Bromine Iron and Fluorine 6. 7. 8. 9. 10. Aluminum and Sulfur Nitrogen and Oxygen Nitrogen and Sulfur Lead and Tellurium Hydrogen and Fluorine NAMING IONIC COMPOUNDS (p 56) Tonight we will practice naming ionic compounds (those with a metal and a non-metal.) This is fairly easy. All you do is take the name of the metal, add a space and then add the name of the non-metal with the suffix “-ide”. Then I want you to use the Criss-Cross method to determine its formula (Show work) Example: Sodium + Chlorine make Sodium Chloride Magnesium + Oxygen make Magnesium Oxide Note: For most non-metals, you use all the words except the last syllable. Exceptions are OxygenOxide, NitrogenNitride, and PhosphorusPhosphide. Try also to figure out the formula using the Criss Cross Method. Elements Sodium + Fluorine Barium + Oxygen Aluminum + Nitrogen Potassium + Sulfur Rubidium + Phosphorus Strontium + Iodine Cesium + Fluorine Radium + Arsenic Lithium + Bromine Beryllium + Chlorine Name Steps: 1) Write the Symbols of the Elements, Metal First 2) Put the Oxidation Numbers on Top of the Symbols 3) Criss Cross (Metal gets non-metal's number - Non-metal gets metal's) 4) Reduce and Remove Ones Formula





![[RH] Chemistry - Hodder Education](http://s3.studylib.net/store/data/009655449_1-244c3ac98c70ac57156679dff49dd5a0-300x300.png)