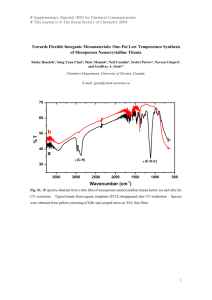
Needs for process engineering in Green and Environmental
advertisement

NEEDS FOR PROCESS ENGINEERING IN GREEN AND ENVIRONMENTAL PHOTOCATALYSIS HERRMANN Jean-Marie *Deputy-Director of IRCELYON, UMR CNRS 5256, Université Lyon1 ; Head of Sustainable Chemistry Cluster of Rhône-Alpes Region ; 2 av. Einstein, 69626 Villeurbanne cedex (France) jean-marie.herrmann@ircelyon.univ-lyon1.fr Heterogeneous photocatalysis is simultaneously able to be efficient in Green Chemistry, in Fine Chemicals and in emerging “Advanced Oxidation Processes” (AOP). 1. Principle of photocatalysis: When a divided semiconductor is illuminated with photons of energy higher than or equal to its band gap energy, photo-electrons and photo-holes are created and produce redox reactions. Photocatalysis is based on the double aptitude of the photocatalyst to simultaneously adsorb reactants and to absorb efficient photons. 2. Green Chemistry and Fine Chemicals: Example of a green photocatalytic reaction. In dry media, photocatalysis is able to induce mild and (regio-)selective oxidations owing to the creation of a neutral atomic O* species. An important intermediate in industrial fine (perfume) chemistry (4-tert-butyl-benzaldehyde) is environmentally-hostilely prepared in industry by a stoichiometric oxidation of 4-tert-butyl-toluene (4-TBT) by permanganate with a lot of (in-)organic by-products: 4-tert-C4H9 -C6H4-CH3+ 4 KMnO4 + 6 H2SO4 4-tert–C4H9–C6H4-CHO + 2 K2SO4+11 H2O By contrast, the oxidation of 4-TBT is 100% selective, both in gas or liquid phase, in 4-tertbutyl-benzaldehyde by mere irradiation of titania at room temperature: 4-tert - C4H9 -C6H4 CH3 + O2 4-tert - C4H9 - C6H4 – CHO + H2O The photocatalytic oxidation of 4-TBT is a typical example of “Green Chemistry” with the use of air and a cheap, stable and recyclable titania catalyst which does not need solvents nor heating but only UV-A light provided by lamps whose technology is permanently improving. 3. Environmental Photocatalysis Titania becomes a total oxidation catalyst once in water or humid air because of the photogeneration of OH° radicals (2nd best oxidant after fluorine) by neutralization of OH surface groups by positive holes h+. 3.1. Water decontamination is obtained by oxidization of most of toxic inorganic ions in their harmless upper oxidization state. Simultaneously, organic pollutants (solvents, pesticides, herbicides, insecticides, fungicides, dyes, etc...) are totally mineralized as evidenced by carbon, nitrogen and hetero-atoms mass balance. 3.2. Air treatment concerns the degradation of VOC’s, and odors. This is applied to clean air in confined atmospheres (clean rooms, refrigerators, hospitals,) on fixed catalytic beds. 3.3. Disinfection: Photocatalysis is also active in the “bio-world” by killing bacteria (E.Coli, streptococcus faecalis,…) in water without re-growth. Virus H5N2, a model virus close to H5N1, responsible for the avian flu, was inactivated in a contaminated air flow of 40 m 3/h with a 99.93% efficiency for a total air disinfection in a single pass. 4-Solar Energy and Applications: The UV-A solar spectrum can be advantageously used for outside applications as demonstrated by solar prototypes in agricultural, water and food industries, especially for removal of odors and self-cleaning properties of outdoor materials. 5- Needs for Green Process Engineering. The present challenge is the development of the photocatalytic engineering for optimization of the reactions, which are performed in a wide range of media and conditions. One has to take into account the polyphasic nature of the systems, including UV-light, the “electromagnetic phase”. Air treatment requires a fixed bed. It is the same for water, to avoid the final tedious filtration. However, it has been shown that disinfection is more efficient with titania slurries since micro-organism cells are ca. 2 orders of magnitude bigger than catalyst particles or their agglomerates. Up to now, no photoreactors have calculated and designed for green photocatalytic reactions in fine selective chemistry.