Notes on the Kinetic-Molecular Theory of Gases
advertisement

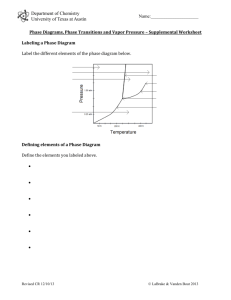
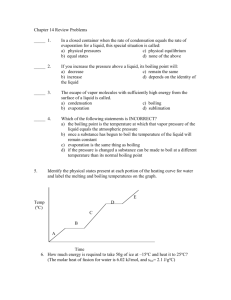
Notes on the Changes of State & Phases Name Describe what it takes to be in a phase Equilibrium exists only when … Le Châtelier’s principle states that if a system at equilibrium is subjected to stress, the equilibrium is shifted in the direction that tends to relieve the stress. Explain how a glass of ice-water sitting in the hot summer sun illustrates equilibrium Vapor pressure exists in a can of un-opened soda pop especially after it has been shaken or warmed. What is vapor pressure? Why do you need a liquid to have vapor pressure? List factors that effect the shift of equilibrium between the evaporation and condensation of molecules in a closed container. Favoring Evaporation (Gas molecules) Favoring Condensation (Liquid molecules) From the Visual Concepts Demo From the Visual Concepts Demo List the factors What are the three main factors that effect equilibrium? Explain the effect What do Acetone (Nail Polish Remover), Gasoline, and Sweat all have in common? … They are all Volatile liquids. A Volatile liquid is different from non-volatile liquid because the volatile liquid will …. Is Evaporation the same as Boiling? Explain. A Boiling Curve for water is a graph comparing which two variables? _____________ ___________ On a Phase Diagram for Water the Normal Boiling Point of H2O exists at a pressure of ____ Atm & ____ C On a Phase Diagram for Water the Normal Freezing Point of H2O exists at a pressure of ___ Atm & ____ C Pressure in Torr Diethyl ether 200 Ethanol 200 Water 200 The six possible Changes of State Name of Phase change Original Phase New Phase Real life example 1) 2) 3) 4) 5) 6) Using the Boiling Curve on page 344 of your book determine the temperature at which the following molecules will boil as the pressure changes. Molecule Define the following (Pg 347) Phase Diagram Triple Point Critical Point Critical Temperature Critical Pressure Use the phase diagram for water in your book to complete the table.(pg 389) a) b) c) d) e) f) g) Pressure (atm) 0.0045 0.870 125.0 90.5 0.0020 200.0 Temperature (C) 0.005 15.5 –1.5 385.0 125.0 100.1 Phase Vapor (gas) Liquid Solid Boiling Temperature