PTTa - Leek High School
advertisement

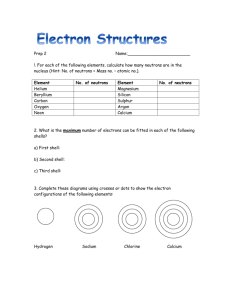
Group 1 (alkali metals) elements - reactivity Group 1 elements (alkali metals) were first discovered from 1807 onwards. Some of the physical properties are shown below. Element Boiling point oC Lithium 1342 Sodium 883 Potassium 760 Rubidium 686 Caesium 670 Melting point oC 181 98 63 39 29 Density g/cm3 Atomic mass Diameter of atom (nano-metres) 0.53 0.97 0.86 1.54 1.88 3.00 0.246 23 0.304 39.1 0.406 85.5 0.432 132.9 0.470 As the atomic mass (and the size of the atom) of the Group 1 alkali metals increases the: boiling point melting point all decrease whilst the density increases. The reason why these alkali metals are reactive is that they all have one electron in their outer shell. This means that the atoms of the alkali metals are not "very happy" and they will do all they can to lose their one lone outer electron In order of reactivity caesium is the most reactive followed by rubidium, then potassium, sodium and lithium is the least reactive. Electronic configurations. Element Atomic number Electrons in 2nd shell 1 Electrons in 3rd shell 3 Electrons in 1st shell 2 Lithium Electrons in 4th shell Electrons in 5th shell Sodium 11 2 8 1 Potassium 19 2 8 8 1 Rubidium 37 2 8 18 8 1 Caesium 55 2 8 18 18 8 Electrons in 6th shell 1 Electronic configurations (diagrams) for lithium, sodium and potassium are shown below. NOTE As the atom becomes bigger and bigger the electron in the outer shell is held less tightly and is more likely to escape. So for caesium (size 0.470 nm) the outer electron is not held tightly. It can easily be lost. Caesium IS very reactive. So for lithium (size 0.246 nm) the outer electron is held tightly. It is hard to lose. Lithium is NOT very reactive. As these metals are so reactive they are stored: in an inert atmosphere (caesium and rubidium) under oil (lithium, sodium and potassium)