Physical properties of transition metals and their
advertisement

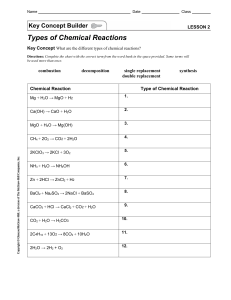
Decomposition Reactions Try balancing the following equations on your own Note: You must type a number into the box , even if it is 1. Use whole numbers use the lowest common multiples i.e if the numbers 2,2,4 make the equation balanced - this will be marked wrong as the numbers 1,1 and 2 are in the simplest whole number ratios) 1CaCO3CaO+CO2 2MgCO3MgO+CO2 3ZnCO3ZnO+CO2 4AgClCl2+Ag 5Ca(OH)2CaO+H2O 6Al(OH)3Al2O3+H2O 7NaNO3NaNO2+O2 Harder examples of decomposition 8Pb(NO3)2PbO+NO2+O2 9NaHCO3Na2CO3+H2O+CO2 10FeSO4Fe2O3+SO3+SO2