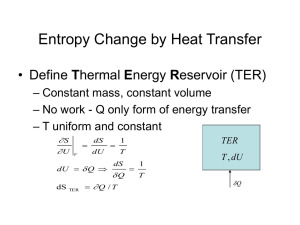
The Carnot Cycle
The most efficient cycle is one consisting solely of ideal
reversible processes. The Carnot cycle is such a cycle
and it provides an upper limit on the performance of a
real cycle operating between the same two thermal
reservoirs.
Carnot cycle consists of the following 4 reversible
processes:
12 Adiabatic compression
23 Isothermal expansion
34 Adiabatic expansion
41 Isothermal compression
TCTH
THTH
THTC
TCTC
120
Process 1-2 Reversible Adiabatic Compression:
- Gas is compressed very slowly (quasi-equilibrium) so
pressure remains uniform throughout the system
- Work done on the system:
U = Q – (-W) U = +W
- Gas temperature increases from TC to TH
Process 2-3 Reversible Isothermal Expansion
- Gas expands slowly, gas cools but as soon as the
temperature drops dT it obtains heat from hot reservoir
raising the temperature back to TH. Since temperature
difference is always dT heat transfer is reversible
- Work done by the system
U = QH – (+W) QH = +W
- Heat transfer QH to the gas, temperature stays at TH
Process 3-4 Reversible Adiabatic Expansion:
- Gas expands very slowly (quasi-equilibrium) so pressure
remains uniform throughout the system
- Work done by the system:
U = Q – (+W) U = -W
- Gas temperature decreases from TH to TC
Process 4-1 Reversible Isothermal Compression
- Work done on the system
U = QC – (-W) QC = -W
- Heat transfer QC from the gas, temperature stays at TC
121
Carnot cycle on a P-v diagram
QH
TH
QH
Wnet
E
QC
TL
QC
Net work per cycle per unit mass= Pdv
Wcycle
(-)
(+)
(+)
(-)
12 Pdv 23 Pdv 34 Pdv 41 Pdv
shaded area (net work out)
Net heat transfer per cycle = QH - QC
122
Carnot Refrigeration Cycle
The Carnot heat-engine cycle is composed of totally
reversible processes. The reverse of the heat engine cycle
is the Carnot refrigeration cycle.
4
QH
TH
QH
3
Wnet
E
QC
TL
1
QC
2
Heat is removed from the cold reservoir and heat is added
to the hot reservoir.
Wcycle
(+)
(-)
(-)
(+)
12 Pdv 23 Pdv 34 Pdv 41 Pdv
shaded area (net work in)
123
Carnot Principles
1. The efficiency of an irreversible heat engine is always
less than the efficiency of a reversible one operating
between the same two reservoirs.
2. The efficiencies of all reversible heat engines operating
between the same two reservoirs are the same.
Violation of either principle results in the violation of K-P
Example: Consider two heat engines operating between
the same two reservoirs, one reversible and the other
irreversible, and each engine is supplied the same amount
of heat QH
TH
QH
WI
QH
I
R
QC,I
WR
QC,R
TL
In violation of Principle #1 assume ηI > ηR,
since
W
WI > W R
QH
124
Run reversible engine as a refrigerator
TH
QH
QH
WI
I
WR
R
QC,I
QC,R
TL
Heat rejected from refrigerator goes to the irreversible
engine
QH
QH
WI
I
WR
R
QC,I
QC,R
TL
The combined engine is:
C
WI - WR
QC,R – QC,I
TL
The combined engine draws heat from a single reservoir
and generates net work WI –WR violates K-P statement
125
Kelvin Temperature Scale
Carnot Pr. #2 implies that the efficiency of a reversible
cycle is independent of the working fluid and depends
only on the temperature of the reservoir ηR = g(TH,TC)
Recall : cycle 1
QC
Q
C f (TH , TC ) need to find f
QH
QH
Consider three reversible engines A, B and D. Where A
and D draw heat Q1 from the same reservoir at T1
T1
Q1
Combined
reversible
cycle “C”
WA
T2
WB
Q1
A
Q2
D
B
WC
Q4
Q3
T3
Engines A and B can be combined into one reversible
engine C
According to Carnot Pr. #2
So, 1
ηC = ηD
Q1
Q1
therefore Q3 = Q4
1
Q3
Q4
126
For each engine we can state (Q4 = Q3)
Q1
f (T1 , T2 )
Q2
Note,
Q2
f (T2 , T3 )
Q3
Q1 Q1
f (T1 , T3 )
Q4 Q3
Q1 Q1 Q2
f (T1 , T3 ) f (T1 , T2 ) f (T2 , T3 )
Q3 Q2 Q3
Since left hand side (LHS) is independent of T2 the RHS
must also be independent of T2
This condition is satisfied only if:
f (T1 , T2 )
(T1 )
(T2 )
and f (T2 , T3 )
(T2 )
(T3 )
Q1 (T1 ) (T2 ) (T1 )
Q3 (T2 ) (T3 ) (T3 )
Therefore, for a reversible heat engine operating between
two reservoirs at temperature TH and TC
QH (TH )
QC (TC )
127
Lord Kelvin proposed taking (T) = T, so that
QH
QC
T
H
rev TC
TH
QH
R
Wcycle
QC
TC
The thermal efficiency of a reversible heat engine is
Carnot
engine
QC
TC
1
1
QH
TH
The thermal efficiency of a reversible refrigerator is
Carnot
refrig
1
QH
1
QC
1
TH
1
TC
This is known as the Carnot Efficiency, it represents the
highest possible efficiency for an engine operating
between two reservoirs at TC and TH
128
Use reversible heat engine to measure temperature of an
object at temperature T on Kelvin scale by measuring QH
and QC
T
QH
R
W
QC
Use water at its triple
point 273.16K in the
lower reservoir
273.16K
Measure QH and QC to get the temperature of reservoir T
e.g.,
QH
T TC
QC
QH
QC
273.16K
129
 0
0