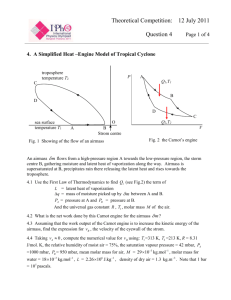
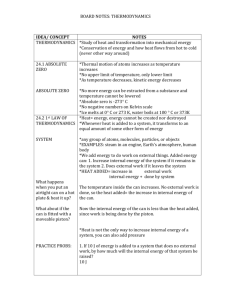
PV Diagrams
advertisement

AP Physics – 1st Law Continued 1. Collect and read the handout on Heat Engine Efficiency from the front of the room 2. When finished work your way through the worksheet provided 3. In 20 minutes, we will explore PV diagrams and the Carnot Cycle 4. Next Class: The 2nd Law of Thermodynamics PV-Diagrams Pressure/Volume graphs are of tremendous value in analyzing the performance of heat engines. Essentially, the amount of work produced by an engine depends on the path it takes (changes in pressure and volume) PV-Diagrams Confused…? Expected, let’s start slow 1. Engines follow a path, in this case from 1 2, 23, 34, and 41 2. Each affects the Work done by, or done on the System Net Work of a Machine Let’s study the engine to the right… P Q: is the direction of the arrows P2 important? P1 c d a b V1 V2 V Net Work of a Machine Let’s study the engine to the right… Q: is the direction of the arrows important? YES! Challenge is if work is done ON the system or BY the system Note: Please write down the work done for each of the next for steps P P2 P1 c d a b V1 V2 V Step 1: Process a b represents an isobaric compression of a gas From a to b, the pressure remains constant – its value is P1. P The volume decrease from V1 to V2. This represents work done on the system. How can you calculate the W? P1 a b V1 V2 V Step 1: Process a b represents an isobaric compression of a gas From a to b, the pressure remains constant – its value is P1. P The volume decrease from V1 to V2. This represents work done on the system. How can you calculate the W? W = PDV ( = P1 V1 -V2 ) P1 a b V1 V2 V Step 2: Process b c is an isochoric compression Isochoric because the volume does not change but the pressure increases. What is the work for this step? P P2 c 0!!! P1 b V1 V Step 3: Process c d is an isobaric expansion. The gas expands from V1 to V2, doing work as it expands. The amount of work is equal to the area under the curve. W = PDV ( = P2 V2 -V1 P P2 c d ) V1 V2 V Step 4: Process d a is an isochoric expansion Volume stays constant. So no work is done. Pressure decreases. W = PDV () =P 0 =0 P P2 P1 c d a b V1 V2 V Complete Cycle Based on what you have recorded for the work on each step, what is P the total work done on the system? P2 P1 c d a b V1 V2 V Complete Cycle Based on what you have recorded for the work on each step, what is P the total work done on the system? P2 ( W = P2 V2 -V1 ) ( - P1 V1 -V2 c d ) OR just the area of the enclosed area! P1 a b V1 V2 V Whiteboarding! 1. Form a group of 3 or 4 no more no less 2. Collect a Whiteboard and pens, and work your way through the following two problems 3. Make sure you are contributing to your group, if you are not then start a new group! Question 1: A substance undergoes a cyclic process shown in the graph. Heat transfer occurs during each process in the cycle. (a) What is the work output during process a b? P ( atm) a 5 .0 0 (b) How much work input is required during process b c? 1.0 0 c (c) What is the net work done during the cycle 10 .0 Note: 1 atm = 1.013 x 105 Pa Note: 1 L = 0.001 m-3 b 5 0 .0 V (L) Question 1: A substance undergoes a cyclic process shown in the graph. Heat transfer occurs during each process in the cycle. (a) 1.22 x 104 J (Expansion process, so work is done on surroundings) P ( atm) a 5 .0 0 (b) -4.05 x 103 J (c) -8.15 x 103 J 1.0 0 c 10 .0 b 5 0 .0 V (L) Question 1: Question 2: A heat engine’s cycle is shown in the PV diagram to the right. P P1 = 345 kPa, P2 = 245 kPa, P3 = 125 P1 k Pa, and P4 = 225 kPa. V1 = 35.0 L P2 and V2 = 85.0 L. What is the net work done during one cycle of the engine? P4 P3 a b d c Note: 1 L = 0.001 m3 V1 V2 V Question 2: A heat engine’s cycle is shown in the PV diagram to the right. P1 = 345 kPa, P2 = 245 kPa, P3 = 125 k Pa, and P4 = 225 kPa. V1 = 35.0 L and V2 = 85.0 L. What is the net work done during one cycle of the engine? P P1 a P2 P4 P3 b d c Note: 1 L = 0.001 m3 Ans: 6.00 kJ V1 V2 V Question 2: 1st Law + Carnot Cycle – An Ideal Engine 1. Please put away the whiteboards 2. Collect the 1st Law Up to Now Worksheet + the handout on the Carnot Cycle, make sure to read this! 3. By now you should have complete most of the MC questions from the Thermo Package 4. We will have our last quiz in 20 minutes! 1st Law incomplete • Conservation of Energy, however processes that do conserve energy, still don’t appear to happen! WHY!? A coffee cup breaks spontaneously but is unable to get put back together spontaneously Clausius statement: “it is impossible for a self acting machine working in a cyclic process without any external force, to transfer heat from a body at a lower temperature to a body at a higher temperature.” Engine Efficiency “Engine Statement” It is impossible for any system to undergo a process in which it absorbs heat from a reservoir at a single temperature and converts the heat completely into mechanical work, with the system ending in the same state in which it begins. Workless Refrigerator “Refrigerator Statement”: It is impossible for any process to have as its sole results the transfer of heat from a cooler to a hotter body 2nd Law of Thermodynamics Efficiency ≠ 100% The Carnot Cycle represents the MAXIMUM efficiency for a heat engine 1. Isothermal exp. 2. Adiabatic exp. 3. Isothermal comp. 4. Adiabatic comp. Efficiency of Carnot Cycle TH - TC ecarnot = x100% TH eheat engine £ ecarnot Quiz! ~ Heat Engines Please set up for 4th and final quiz More Handouts! • Please collect three more handouts on Efficiency of Engines, the Second Law and… • AP Thermodynamics Wrap-up that reviews all the equations and how the work… VERY USEFUL! • Next Class: Thermo Unit Test AP Physics Tutorial: Next Tuesday Morning at 7:30-8:15 AM Please bring questions! If you’re at home we will be doing a Socrative Review! Please log in and take it!