7 | Energy and Chemical Change 253 7.1 Energy: The Ability to Do
advertisement

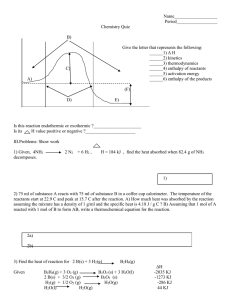
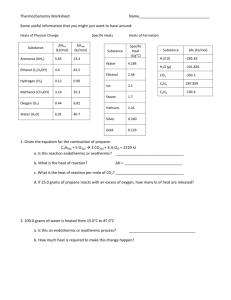
7 | Energy and Chemical Change 253 7.1 Energy: The Ability to Do Work 254 7.3 Measuring Heat 259 7.5 Heat, Work, and the First Law of Thermodynamics 267 7.7 Thermochemical Equations 275 7.9 Standard Heats of Reaction 283 for water : 7.2 Internal Energy 257 7.4 Energy of Chemical Reactions 265 7.6 Heats of Reaction 270 7.8 Hess’s Law 277 heat of fusion = 333 J / g heat of vaporization = 2260 J / g specific heat capacity (s) = 2.1 J / g K (l) = 4.2 J / g K (g) = 2.0 J / g K 1. What is the amount of heat needed to convert 350.g of ice at -35.0C to -11.0C? 2. What is the amount of energy needed to completely melt 200.0g of ice sample starting at -22.0C? 3. What is the water sample size if 296J is released when this water sample cools from 421C to 322C? 4. The T of a block of metal (70.0g) went from 35.7C to 100.2C when 579J of energy was added. What is the identity of the metal if pure? 5. A metal spoon (18.9 J/gC, 3.5g) at 25C was placed in 300. mL of water at 65C. What is the final temperature? 6. Write an equation that goes with Hf forH2O(l)? C3H8(g)? CO2(g)? 7. Hf (kJ/mol) for H2O(l) (-285.9) ; C3H8(g) (-104) ; CO2(g) (-393.5) Using the enthalpy of formation, what is H for this reaction? (a) (b) 8. 2H2(g) C3H8 (g) + + O2(g) 5O2(g) H2O (g) ---> H2(g) + 1/2 O2(g) ---> --> 2H2O (l) 3CO2(g) + 4H2O(l) H = +241.8kJ a) H given here above called “ H for the reaction’, what else can it be called? b) Calculate H for H2(g) + 1/2 O2(g) -- H2O (g) c) H given in (b) is called H for the reaction, what else can it be called? d) What is the enthalpy change for decomposition of 38.9g of water(g) using the equation given? e) What is the enthalpy change for formation of 138.g of water(g) using the equation given? 9. A sample of glucose weighing 5.00g was burned in a bomb calorimeter containing 2.00L of water. The temperature of the water and the calorimeter rose by 2.4C. Calculate the heat of combustion of glucose, C6H12O6 (180.0g/mol) in kJ/mol. The heat capacity of the calorimeter reads 26J/C. 10. (a) Using the information below, what is the H for N2(g) + 2 O2(g) --> 2NO2(g) ? NO(g) + ½ O2(g) ---> NO2(g) 2 NO(g) ---> O2(g) + N2(g) H H = -56kJ = -180.kJ (b) Using the information below, what is the H of formation for H2O(l)? 2 H2O (g) ---> 2 H2(g) + O2(g) H = + 482kJ H2O (g) ---> H2O (l) H = -44kJ