enthalpy changes calculations
advertisement

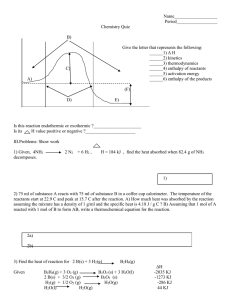
Enthalpy change calculations 1) When gaseous hydrogen and oxygen are heated in a test tube, droplets of liquid water form on the sides of the test tube. Calculate f H (H2O(ℓ)), given the following data: f H (H2O(g)) = – 242 kJ mol–1 vap H (H2O(ℓ)) = + 44 kJ mol–1 2) i) Ammonia can be oxidised to produce nitrogen, N2, and steam as shown in the equation below: 4NH3(g) + 3O2(g) 2N2(g) + 6H2O(g) D r H = –1267 kJ mol–1 Calculate the energy produced when 50.0 g of ammonia reacts as shown in the equation above. 3) fH (HBr, g) is –36.2 kJ mol–1. Calculate the heat produced by the formation of 50.0 g of HBr(g) from its elements in their standard states. 4) The experimental value for fH(H2O, ℓ) is –286 kJ mol–1 and the enthalpy of formation of water in the gas state, fH (H2O, g) is –241 kJ mol–1 Using the information, calculate the vapH(H2O), and also the heat required to vaporise 100 g of water. © 2014 http://www.chemicalminds.wikispaces.com NCEA questions and answers reproduced with permission from NZ