27c991fbdcbf6ad4608e42b666974123
advertisement

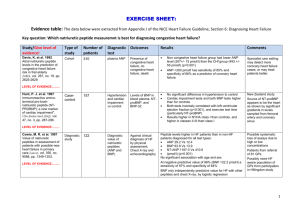
Widespread Targeted Review Results Part A – CPT 83880 TOB 14X Medical Review Part A has recently completed the widespread targeted review for Q2 2014 of CPT 83880, Pathology and Laboratory: B-type Natriuretic Peptide (BNP) Testing for Type of Bill 14X. The claims were randomly selected across the provider community billing this service that met the parameters of the edit. The topic code for this review was 5134K for Alabama providers, 5010R for Georgia providers and 5004N for Tennessee providers. Based on the results of this review the widespread targeted review will be continued until the error rate is decreased. State Alabama Georgia Tennessee CPT 83880 Number of Providers 62 81 69 Error Rate 100% 100% 99% The majority of the denials were due to one of the following reasons: 1. Lack of medical necessity to support providing the service for 83880 (Denial Reason 54155, 5T013): This denial reason was due to the documentation submitted did not support clinical urgency for 83880. The lab was ordered routine. This service is considered reasonable and necessary for establishing the diagnosis of CHF in acutely ill patients presenting with dyspnea or for predicting the long term risk of cardiac events or death across the spectrum of acute coronary syndromes when measured in the first few days after an acute event. In some instances the patient already had an established diagnosis of CHF. 2. Lack of documentation (Denial Reason 55520/5T011): Claims were denied due to the lack of documentation to review for services provided on the claim. Claims either did not include physician order, laboratory results or progress notes to support the service was provided as submitted on the claim. 3. Lack of timely submission of requested documentation (Denial Reason 56900): Claims were denied due to a lack of record submission in a timely manner. According to The Medicare Program Integrity Manual, PUB 100-8, Chapter 3, §3.2.3.8b, “ During prepayment… or post payment… review, if no response is received within 45 calendar days after the date of the ADR, the MACs, and ZPICs shall deny the claim.” To eliminate 56900 denials, please review the following elements to ensure appropriate and timely record processing: Print screen the ADR letter and attach the copy to the front of the requested medical documentation Send requested information to the address on the ADR letter Do NOT send certified mail or overnight mail Include ALL requested documentation outlined in the ADR Submit the above information via regular mail in a timeframe to ensure the MAC receives the information by the 45th day after the date of the request. The medical review decisions were based on the LCD for Pathology and Laboratory: B-type Natriuretic Peptide (BNP) Testing (L30046) can be found at: https://www.cahabagba.com/part_a/policies_medical_review/lcd_active.htm. Please review the following information from Local Coverage Determination L30046: Indications: 1. Congestive Heart Failure (CHF) is a complex clinical syndrome characterized by dysfunction of the left, right, or both ventricles, which results in the impairment of the heart’s ability to circulate blood at the rate sufficient to maintain the metabolic needs of the peripheral tissues and various organs. B-type natriuretic peptide (BNP) is synthesized, stored, and released primarily by the ventricular myocardium in response to volume expansion and pressure overload, which are hemodynamic parameters in CHF. Used in conjunction with other clinical information, serum BNP concentrations parallel dyspnea in heart failure suggesting its usefulness as a neurohormonal index of progressive heart failure. Serum BNP, for the purposes of coverage, is considered a ‘point-of-service test (performed and immediately used in the disposition of patient care). 2. Serum BNP, when used in conjunction with other clinical information, will be considered reasonable and necessary for the following: A. Establishing the diagnosis of CHF in acutely ill patients presenting with dyspnea. B. Predicting the long term risk of cardiac events or death across the spectrum of acute coronary syndromes when measured in the first few days after an acute coronary event. Since this situation is an inpatient service, it is not addressed in this LCD. Limitations 1. Serum BNP will be considered noncovered in the following: A. Monitoring the efficiency of treatment for CHF B. Tailoring the therapy for heart failure 2. Since BNP is a point of service test, the primary outpatient site of service expected to perform a serum BNP is the emergency room or a physician’s office. References: Cahaba Local Coverage Determination (LCD) for Pathology and Laboratory: B-type Natriuretic Peptide (BNP) Testing (L30046) Title XVIII, Social Security Act, Section 1833(e) Title XVIII, Social Security Act, Section 1862(a)(1)(A)