2dOsmosis Dialysis Tubing Lab
advertisement

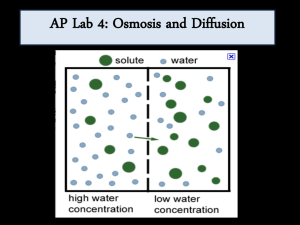
Biology 30S Name Investigating the Movement of Starch, Iodine, and Glucose Across a Selectively Permeable Membrane Scientific Method 1. Make observations about the natural world. 2. Ask questions about those observations. 3. Formulate a reasonable testable hypothesis to explain observations. 4. Create, execute, and replicate experiments testing the hypothesis by generating data. 5. Analyze data in comparison to hypothesis and draw inferences. This stimulates further inquiry. The cycle begins anew. Purpose We will divide this lab into three parts: Part 1 – To explore how different molecules move by diffusion and osmosis through semi-permeable membranes. Part 2 – To understand how different concentration gradients affect the movement of substances through semipermeable membranes. Part 3 – To understand how the processes of diffusion and osmosis through semi-permeable membranes contribute to homeostasis in single-celled, aquatic organisms Background & Key Terms All living cells are surrounded by lipid membranes. These lipid membranes are semi-permeable, permeable to some molecules, and not others, which are determined by molecular size and charge. This semi-permeability controls diffusion and osmosis. In this lab, we will examine the property of selective permeability utilizing dialysis tubing (a specialized tubing that sorts by size), water, and a set of solutions designed to reproduce situations cells are faced with. Starch molecules are considerably larger that both glucose and iodine molecules. 1. Diffusion: The movement of a substance from a region of high concentration to a region of low concentration. 2. Solute: A substance that is dissolved in a solution. 3. Solution: A homogeneous, liquid mixture of two or more substances. The relative solute concentrations between two solutions are compared as: a. Hypotonic solution: A solution with a lower solute concentration than another solution. b. Hypertonic solution: A solution with a higher solute concentration than another solution. c. Isotonic solutions: Solutions of equal solute concentration. 4. Osmosis: The diffusion of water across a semi-permeable membrane. 5. Semi-permeable Membrane: A membrane that allows some molecules, but not others, to pass through it. 6. Homeostasis: The inherent tendency in an organism to maintain physiological balance. Questions: 1. How do semi-permeable membranes and the processes of diffusion and osmosis contribute to homeostasis in single-celled, aquatic organisms? 2. How does the molecular size of a solute affect these processes? 3. How does the concentration of solute affect the rate of diffusion and osmosis? 4. What happens if one solute can pass through the membrane, but another can’t? Part 1 – Molecules and Diffusion/Osmosis through a Semipermeable Membrane Hypothesis Materials Beakers (250 ml) Potassium iodide solution (IKI) Glucose Test Strips Dialysis tubing Glucose solution Molasses (sucrose) solution Starch solution Distilled water String Scissors 5 6 drops (3 drops/beaker) 3 5 – 20 cm strips 15 ml 500 ml 500 ml 500 ml 10 – 20 cm pieces 1 pair Method We will utilize solutions of starch, glucose, sucrose (molasses), and iodine to test these hypotheses using dialysis tubing as a model of the membrane of cells. Below, we lay out basic techniques that you can use to structure experimental protocols. Before we start, we should mention a little chemistry. When glucose or starch is dissolved in water they are colorless (or milky white). Starch and iodine react when in solution and turn blue in color. Additionally, we can test for the presence of glucose in solution with a urinalysis test strip. These will help you to determine what solutes are passing through the semi-permeable membrane. You will set up an experiment where you fill dialysis tubes and beakers as outlined below. Think about this experimental design, and make predictions about what results you might expect to see from your experiment, based on the hypothesis you are testing. Be sure to record your observations in Table 2 below. • Test #1: Cut a piece of dialysis tubing of approximately 20 cm in length. Tie one end of the tubing and fill it with a mixture of water and molasses. Tie the other end of the tubing and place it into a beaker of water. • Test #2: Cut another piece of dialysis tubing and tie one end. This time, pour water into the tubing. Tie the other end of the tubing and place it into a beaker containing a mixture of water and molasses. • Test #3: Place a dilute iodine solution into a piece of dialysis tubing. Tie the other end of the tubing and place it into a beaker containing a starch and water mixture. • Test #4: Pour a starch and water mixture into a piece of dialysis tubing. Place the tubing into a beaker containing a dilute iodine solution. • Test #5: Pour a glucose solution into a piece of dialysis tubing. Use one glucose test strip to test for the presence of glucose in the solution you have just put into the dialysis tube. At the end of the class, place a glucose test strip into a beaker containing water to test for the presence of glucose. Repeat this test the following day. 2 Results/Observations Describe the appearance of each solution or mixture before the procedure. Record detailed observations at the end of the class as well as the following day. Your results can be recorded in the table below: Table 2 Observations Tubing Beaker Initial -tubing Test #1 molasses & water water Test #2 water molasses & water Test #3 Iodine solution starch & water Test #4 starch & water Iodine solution Test #5 glucose solution water -beaker End of Class -tubing -beaker Following Day -tubing -beaker Analysis Thought questions: 1. If the dialysis tubing represents a human body cell, how would your results affect how a human body cell maintains homeostasis? 2. What do the contents of the beaker and the tubing represent in the human body? BeakerTubing3. For each of the procedures, indicate which molecules cross the membranes and which molecules do not. Explain how you came to this conclusion. Use the following terms to explain why each type of molecule is capable or incapable of crossing the membrane and how the movement takes place: semi-permeable membrane, concentration, hypertonic, hypotonic, &isotonic. Test #1 Test #2 Test #3 Test #4 Test #5 4. Assuming that a molecule is able to pass through the dialysis tubing, is the dialysis tubing a one-way or twoway path? Refer to experimental evidence to support your answer. 3 Conclusion Were your predictions correct? Did you support or reject the hypothesis you were testing? Refer to experimental evidence to support your statements. 4 Part 2 – The Effect of Concentration Gradient on Osmosis Background: You will be working with different concentrations of starch and looking at how they affect diffusion across a semi-permeable membrane, the dialysis tubing. This part of the experiment is not completely written out and relies on you to establish the protocol. Don’t worry; we will give you some hints to get you started. Sucrose (common table sugar) is a molecule composed of one glucose molecule, and one fructose molecule. If a glucose molecule were able to pass through a membrane, would it be safe to assume the sucrose would also? What about the other way around? First, you need to think about how you will be able to quantify the amount of diffusion that occurs between the different concentrations. Table 3 lists the materials that are available for your experiment. Table 3. Part 2 materials available Materials: 0.2 M sucrose solution 0.4 M sucrose solution 0.8 M sucrose solution 1.0 M sucrose solution Dialysis tubing Electronic scale 10 ml pipets 2.0 ml pipets Equipment stands Amount per group: 15ml 15ml 15ml 15ml 2 feet 1 1 4 5 After discussing possible protocols with your group, and teacher, outline the experiment you will conduct to test your hypothesis about how the concentration gradient affects the movement of water through a semi-permeable membrane. (Hint: You will also want to set up a table to record your results and graph your final results in your notebook.) Based on lecture and results from your first experiment, formulate an explicit prediction(s) about what you expect to have happen in this experiment. Write it down here: 6 Part 3: Real cell membranes are lipid bilayers that we believe will block the movement of charged ions like sodium and calcium. Water can pass through cell membranes. We will test this hypothesis using table salt (NaCl) and water. The cellular membranes of red onions are semi-permeable, permitting hydrophobic (uncharged) but not hydrophilic (charged) molecules or ions to pass. Sodium ions cannot pass through, but water can. Table 4. Part 3 materials Materials needed: Red Onion Slice Amount needed per group: 1-2 cm 1 1 1 1 Razor Blade Slide Cover Slip Microscope 2 1. Cut a small square out of a layer of the red onion (1-2 cm2). 2. Slice off/peel the thin red layer on the outside of the square using a razor. The thinner the layer, the better, so take your time. 3. Place the sample on a slide and try to keep it from folding over. Place a cover slip over the sample. 4. Place the slide on the microscope and focus to the layer of cells that are filled with red pigment. Try to find an area that you can clearly see individual cells. 5. Draw the cellular structure of these cells in your notes (note your magnification). If you look closely, you can see cell nuclei too. 6. Without removing the slide from the scope, drip a drop or two of 15% NaCl onto the edge of the cover slip. It should be pulled under the coverslip by capillary action, and cover your onion layer. 7. Continue to watch the cells for the next few minutes. You may want to move your slide around to see other areas for the layer since they may not all react the same way. Record your observations. Thought Questions: 8. Was the cell hypo-, hyper-, or isotonic relative to the NaCl solution? Refer to experimental evidence to support your answer. 9. What do you think would happen to a single-celled freshwater organism if it were suddenly thrown in the ocean? Refer to experimental evidence to support your answer. 10. Early life evolved in the sea before moving onto land. What are the challenges faced by a single-celled aquatic organism that tries to live on land? Please explain with reference to the following terms: osmosis, diffusion, solute concentration, and semi-permeable membrane. 7