Diffusion & Osmosis Lab: Cell Membrane Permeability
advertisement

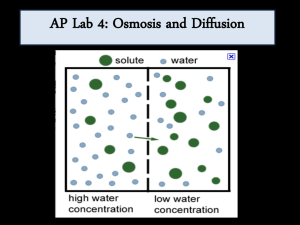
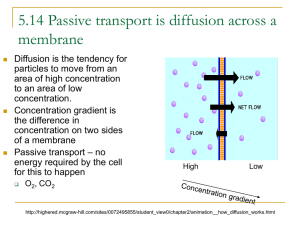
Diffusion and Osmosis OBJECTIVES: 1. To explore how different molecules move by diffusion and osmosis through semi-permeable membranes. 2. To understand how concentration affects the movement of substances through semi-permeable membranes. 3. To understand how the processes of diffusion and osmosis through semipermeable membranes affect homeostasis in Cells I. BACKGROUND MATERIAL All cells are separated from their external environment by a lipid membrane. Cell membranes regulate the molecular traffic in and out of the cell. As a result of their supra-molecular structure (many molecules ordered into a higher level or organization), these membranes exhibit the property of selective permeability. Selective permeability refers to the ability of certain molecules to pass through the membrane while other molecules are blocked by the membrane, or must use special protein pores to pass through the membrane. In this lab, we will examine the property of selective permeability, utilizing dialysis tubing and a set of solutions designed to model the osmotic situations cells handle. NOTE: Since these processes only operate efficiently over short distances, the basic living thing – a cell, is limited in the size it can attain. This is a basic consideration in the evolution of life. TERMS Diffusion: The random movement of molecules or particles, resulting in the net movement of a substance from a region of high concentration to a region of low concentration. Osmosis: The diffusion of water across a semi-permeable membrane. Solute: A substance that is dissolved in a solution. Solution: A homogeneous, liquid mixture of two or more substances. Semi-permeable Membrane: A membrane that allows some molecules, but not others, to pass through it. Homeostasis: The inherent tendency in an organism to maintain physiological balance. 1 II. FORMING HYPOTHESES 1. Make observations about the natural world. 2. Ask questions about those observations. 3. Formulate a reasonable testable hypothesis to explain observations. 4. Collect data to test the hypothesis. 5. Evaluate the hypothesis by comparing it to the collected data. Observation: All living cells are surrounded by lipid membranes. These lipid membranes are semi-permeable. Which type of molecule can pass through the membrane is determined by the types of pore proteins present. This semi-permeability establishes concentration gradients across the membrane, altering diffusion and osmosis rates. Dialysis tubing is a membrane that is selective based on the size of solutes, and can be used as a model for cellular membrane. Questions: How does the molecular size of a solute affect diffusion through the dialysis tubing? How does the concentration of solute affect the rate of diffusion and osmosis? What happens if one solute can pass through the membrane, but another cannot? How do semi-permeable membranes and the processes of diffusion and osmosis contribute to homeostasis in cells? Hypotheses: 1. Large starch molecules are too large to pass through the semi-permeable membrane, but glucose and iodine are small enough to pass through the membrane. 2. The concentration of solute on either side of the membrane has no affect on the rate of osmosis because it does not affect the movement of water. 3. Since diffusion and osmosis are physical properties, they must influence homeostasis in living cells 2 Experiment: We will utilize solutions of starch, glucose, sucrose, and iodine to test hypotheses 1 and 2, using dialysis tubing as a model of the membrane of cells. Below, we lay out basic techniques that you can use to structure experimental protocols. Results: Record your results and compare them to the hypotheses. Do your data support or reject each hypothesis? Do your results differ from/ agree with your classmates? What inferences can you make from your own results and from compiled results? III METHODS Hypothesis 1: Large starch molecules are too large to pass through the semi-permeable membrane, but glucose and iodine are small enough to pass through the membrane. Before you can make predictions about the results of your experiment, you need to know a little chemistry. When glucose or starch is dissolved in water they are colorless (or milky white). Starch and iodine react when in solution and turn a solution blue. Additionally, we can test for the presence of glucose in solution with a urinalysis test strip. In this experiment, you will fill a dialysis tube with starch and glucose and submerge it in a beaker full of water and iodine solution. Think about this experimental design, and make predictions about what results you might expect to see from your experiment, based on the hypothesis you are testing. Be sure to record your observations in Table 2. Predictions: 3 Table 1. Part I Materials Materials needed: Beaker Potassium iodide solution (IKI) Glucose Test Strips Dialysis tubing Glucose/starch solution Clamps Amount needed per group: 1 A few drops 3 125mm 10-15ml 2 Experimental procedure: 1. Fill a beaker about 1/2 full of water and add a several drops of Iodinepotassium iodide (IKI) solution, enough to make the solution yellow. 2. Use a glucose test strip to test for the presence of glucose in the beaker. Dip the strip into the solution, remove immediately, and wait 30 seconds before comparing the color with the scale on the bottle. Record the concentration in Table 2. 3. Take one piece of soaked dialysis tubing, fold the end over on itself about 1”, and then close with a clamp 4. When you are ready to fill the dialysis tube, rub the open end of the tube between your fingers to open it up. Use a pipette to fill the tube with 10-15ml of the glucose/starch solution. 5. Use one glucose test strip to test for the presence of glucose in the solution you have just put into the dialysis tube. 6. Clamp the open end of the dialysis tube and then place it into the beaker of water and iodine. Monitor the experiment for changes. Once the results are apparent, record the color of the liquids in the beaker, and the tube. Repeat the glucose test of the solution in the beaker. Table 2. Data Table for Part I: Beaker Contents Color Start After 30 min. Color Records Beaker Contents Glucose Test Strip of Liquid Contents. Solution In Dialysis Tube Dialysis Tube Glucose Test Color Strip Don’t test 4 Evaluation: Compare your results in table 2 to your predictions for this part of the lab. Do these results cause you to reject your original hypothesis? If your data are not in accord with your predictions, how could you revise the hypothesis to match these results, and what new experiment would you use to test the revised hypothesis? Thought questions: 1. If the dialysis tubing makes a good model for the cell membrane, what do the results of your experiment mean for a cell that needs to maintain homeostasis? 2. It the tube represents a cell, what do the contents of the beaker represent, i.e. the solution outside of the dialysis tubing? 3. If a molecule is able to pass out of the dialysis tubing, do you think it would be able to pass the other way? (In other words, is the dialysis tubing a one-way path?) Describe how you could alter the above experiment to determine if the dialysis tubing is semi-permeable one-way or two-ways. Hypothesis 2: The concentration of solute on either side of the membrane has no effect on the rate of osmosis because it does not effect the movement of water. To test this hypothesis, we have provided you with an experimental apparatus that can be used to quantify (measure) changes in volume that result from the process of osmosis. It consists of a glass pipette, a stopper, some dialysis tube, a beaker, and a ring stand. Your TA will demonstrate how to use this set up. How can this apparatus be used to test a hypothesis that has to do with the rate of osmosis? You also have several solutions of sucrose to use to test this hypothesis. See Table 3 for a list of materials available for this experiment. Sucrose (common table sugar) is a molecule composed of one glucose molecule, and one fructose molecule. 5 Table 3. Materials for testing Hypothesis 2 Materials: Amount per group: 0.2 M sucrose solution 20ml 0.4 M sucrose solution 20ml 0.8 M sucrose solution 20ml 1.6 M sucrose solution 20ml Dialysis tubing 125mm Osmosis apparatus 1 Food coloring As needed Experimental procedure: For this experiment, the procedure is not provided, and relies on you to establish it. Discuss with your group, and the class, the hypothesis you are testing, the materials you have available, and develop an experiment that tests the hypothesis. When an experiment has been agreed upon by the class, you can then develop predictions of the experimental results. Describe your experimental protocol, and predictions below. Experimental Procedure: Prediction(s) for experiment 2: Use the space below to record your results. 6 Evaluation: Do the results of this experiment match your predictions and support your hypothesis? Use experimental evidence to support your answers. If the results do not match your predictions, what revised hypothesis would explain what you did see? How could you test this revised hypothesis? Thought Questions: 1. What do the results of your experiment tell you about how the environment might affect a cell’s ability to maintain homeostasis? 2. How might the concentration of a solute influence the rate of osmosis? 3. Do the results of your experiment tell you anything about the influence of concentration on the rate of diffusion in general? 4. How might you test whether the concentration of a solute alters its rate of diffusion? 5. What keeps the solution from flowing out of the top of the pipette? Hypothesis 3: Since diffusion and osmosis are physical properties, they must influence homeostasis in living cells. To test this hypothesis, you will use cells of red onion, and your own cheek epithelial cells. You have a solution of 15% salt (NaCl), and de-ionized water. The procedure to perform these experiments is provided below. Actual cell membranes have been found to be semi-permeable just like the dialysis membrane you used for the previous experiments. However, they don’t exclude molecules only based on size; the charge of the molecule is also important, permitting hydrophobic (uncharged) but not hydrophilic (charged) molecules or ions to pass. When salt (NaCl) is put in to solution with water, it separates into 2 charged atoms one of Na+ and one of Cl-. 7 For this experiment, you will make slides of both Onion cells, and your cheek epithelial cells. The onion cells will be soaked in 15% salt solution. The cheek cells will be soaked in both de-ionized water (pure water) and 15% salt water. Based on this experimental procedure, list your predictions. Predictions: Table 4. Available materials Materials needed: Red Onion Slice Razor Blade Slides Cover Slip Microscope De-ionized water 15% NaCl solution Tooth picks Amount needed 1-2 cm2 1 3 As needed 1 As needed As needed As needed Experimental procedure: Onion cells 1. Cut a small square out of a layer of the red onion (1-2 cm2). 2. Slice off/peel the thin red layer on the outside of the square using a razor. The thinner the layer, the better, so take your time. 3. Place the sample on a slide and try to keep it from folding over. Place a cover slip over the sample. 4. Place the slide on the microscope and focus on the layer of cells that are filled with red pigment. Try to find an area where you can clearly see individual cells. Make a new slide if you need too! 5. Draw the cellular structure of these cells in your notes (note your magnification). If you look closely, you can see cell nuclei too. 6. Without removing the slide from the scope, drip a drop or two of 15%NaCl onto the edge of the cover slip. It should be pulled under the coverslip by capillary action, and cover your onion layer. 8 7. Continue to watch the cells for the next few minutes. You may want to move your slide around to see other areas of the slide since some parts of the tissue may react differently. Record your observations. Cheek epithelial cells: 1. Take 2 slides. Place a small drop of DI water on one, and a small drop of 15% salt water on the other. 2. Using a toothpick, scrape the inside of your cheek several times. Swirl the tip of the pick in the drop on one slide, and then repeat this for the other slide using a new toothpick. Place a cover slip on each slide and observe under the scope. 3. Make notes about the appearance of the cells and then let the slides sit for 5 minutes and re-examine. Evaluation: Based on the results of these experiments, did you support your prediction(s) ? Did your experiment support the hypothesis being tested? Use experimental evidence to support your answers. Thought Questions: 1. Did the cells gain, loose, or maintain volume in each solution? What does this tell you about the solute concentration in the cell relative to each solution? Refer to experimental evidence to support your answer. 2. What do the results of these experiments tell you about the challenges cells face in trying to maintain homeostasis? 3. What do you think would happen to a single-celled freshwater organism if it were suddenly thrown in the ocean? Refer to experimental evidence to support your answer. 4. Early life evolved in the sea before moving onto land. What are the challenges faced by a single-celled aquatic organism that tries to live on land? Please explain with reference to the following terms: osmosis, diffusion, solute concentration, and semi-permeable membrane. 9