[Date]
[Insurance Company]
Attn: [Contact name, if available]
[Address]
[City, State Zip]
[Fax #]
Patient Name: [Patient’s first and last name]
Policy Number: [Insert Patient's Policy Number]
Group Number: [Insert Patient's Group Number]
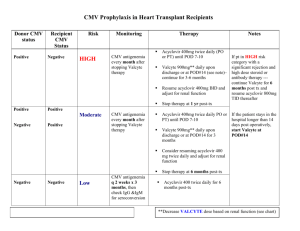
RE: Authorization for Valcyte (valganciclovir HCl)
Please see the enclosed documentation demonstrating the medical necessity of Valcyte (valganciclovir
HCl) for my patient, [patient name]. [He/she] requires prevention of CMV disease subsequent to a [for
adults: kidney/heart/kidney-pancreas; for pediatrics ages 4 months to 16 years: kidney/heart]
transplant. Valcyte tablets were FDA-approved for the prevention of CMV disease in adult kidney, heart,
or kidney-pancreas transplant patients at high risk on September 12, 2003, and for prevention of CMV
disease in kidney or heart transplant patients (4 months to 16 years of age) at high risk on August 28,
2009. I would appreciate prompt review of the information and authorization for Valcyte.
Patient’s Clinical History
[Patient’s name] is a [age]-year-old [male/female] who received a [for adults: kidney/heart/kidneypancreas; for pediatrics ages 4 months to 16 years: kidney/heart] transplant on [date]. [Provide
rationale for prophylaxis. For example, this may include donor and recipient serostatus, brief description
of the patient’s immunosuppressive regimen, and other factors that impact your treatment selection.
Include supporting medical records (see Enclosures section below).]
Rationale
Valcyte (valganciclovir HCl) is an L-valyl ester (prodrug) of ganciclovir that exists as a mixture of two
diastereomers. After oral administration, both diastereomers are rapidly converted to ganciclovir by
intestinal and hepatic esterases. [For adults: Valcyte tablets are indicated for the prevention of CMV
disease in kidney, heart, or kidney-pancreas transplant patients at high risk (Donor CMV
seropositive/Recipient CMV seronegative [D+/R-]). For pediatrics ages 4 months to 16 years: Valcyte for
oral solution and tablets are indicated for the prevention of CMV disease in kidney or heart transplant
patients (4 months to 16 years of age) at high risk (Donor CMV seropositive/Recipient CMV seronegative
[D+/R-]).] It is recommended that prophylaxis start within 10 days of transplantation and continue until
[adult heart or kidney-pancreas, or pediatric heart or kidney transplant: 100 days; adult kidney
transplant: 200 days] post-transplantation.
I would like to treat [patient name] with Valcyte. Please promptly review this information for
authorization of Valcyte for my patient. [He/she] is at risk for CMV disease and requires prophylaxis
beginning within 10 days of [his/her] transplant. I can be reached at [phone number] for additional
information and discussion. Thank you for your prompt attention to this matter.
Sincerely,
[Physician Name]
Enclosures [Suggested]:
Valcyte package insert
Patient clinical/surgical notes & relevant laboratory reports
10537800
 0
0