Intramolecular Forces – Types of Bonding
Electronegativity is a measure of the ability of an atom or molecule to attract electrons in a
chemical bond. The type of bond between two atoms is related to the difference in
electronegativity (∆EN) of the two atoms that are bonded together:
∆EN = EN2 – EN1 , where EN2 is the element with the higher electronegativity
EN1 is the element with the lower electronegativity
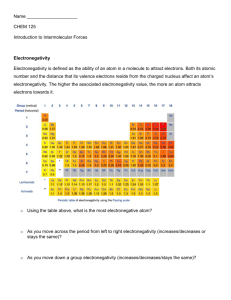
The electronegativity of all the elements can be found on the Electronegativity Periodic Table.
There is a clear periodic trend to electronegativity values – it increase up a group and across a
period.
Let’s look at an example: ammonia (NH3). The nitrogen atom has an electronegativity value of
3.04, while hydrogen has an electronegativity of 2.20. Therefore, the bonded pair of electrons
in any N-H bond has a ∆EN = 0.84.
The greater the ∆EN value, the greater the attraction of an electron pair to the atom of higher
electronegativity (EN2). Although bonding is considered a continuum from pure covalent
character (∆EN = 0) to increasing ionic character, certain ∆EN ranges correspond to one of the
three general types of bonds. See the table below for reference:
∆EN
0.0 to 0.3
0.3 to 1.7
1.7 to 3.3
Type
Covalent (non-polar)
Polar Covalent (polar)
Ionic (very polar)
NON-POLAR
0.0
POLAR
0.3
Description
Equal sharing of electrons
Unequal sharing of electrons
Transfer of electrons
IONIC
1.7
3.3
For our example of ammonia (NH3), ∆EN = ENN – ENH = 3.04 – 2.20 = 0.84. This bond is therefore
classified as a polar covalent bond.
Bond Polarity
In ammonia, the two atoms (nitrogen and hydrogen) have different electronegativities (i.e.,
∆EN > 0). Just like any other polar covalent bond, the two bonding electrons are not equally
shared between the two atoms (nitrogen and hydrogen). Because of this, a bond dipole exists.
The atom that has a greater attraction for electrons (more electronegative) has a partial
negative charge (-), and the atom that has a lower electronegativity has a partial positive
charge (+).
For ammonia, this can be drawn like the following:
Molecular Polarity
The composition and arrangement of the atoms in a molecular structure dictates the shift of
the electron density, and thus, determines the polarity of the molecule. The absence of dipole
moments renders the molecule non-polar. For example, diatomic molecules (i.e., H2, O2, F2, Br2,
I2, N2, Cl2) contain two atoms of the same element; they do not have dipole moments. Binary
molecules - molecules containing atoms of different elements – are polar molecules because
they have a dipole moment.
The following structures show the partial charges and the shift in the electron density for
hydrogen chloride:
However, it is important to note that the presence of polar bonds in a molecule does not
guarantee that the molecule itself will be polar. To determine the polarity of a molecule, you
must find the vector sum of all bond dipoles. If the vectors cancel out, the molecule is nonpolar. If the vectors do not cancel out, the molecule is polar.
The following examples are non-polar molecules. For each vector, there is a vector of equal
proportion that is pulling in an opposite direction. Therefore, the vectors all cancel out.
The following examples are polar molecules. Unlike the previous examples, not all vectors
cancel out.
Notice that both polar molecules and non-polar molecules can have lone electron pairs.
______________________________________________________________________________
Intermolecular Forces of Attraction
The four main types of attractive forces that exist between neighbouring particles are:
1.
2.
3.
4.
London dispersion forces
Dipole-dipole forces
Hydrogen bonding
Ion-dipole forces
These attractions between particles determine the physical properties of substances, which
includes the boiling point, freezing point, vapour pressure, and compressibility.
London Dispersion Forces
-
London dispersion forces of attraction exist between all particles
They are the weakest force of attraction
Are due to the instantaneous dipole-induce dipole attraction
London dispersion forces polarize electrons
The only force of attraction between non-polar molecules; noble gases also exhibit
London forces of attraction
The constant motion of the electrons and their “clouds” will produce momentary dipoles in the
molecule and any neighbouring molecules’ electrons will shift away because of the repulsion
between like charges. The momentary force of attraction is the London dispersion force.
Diagram of how London Dispersion Forces induce dipoles on neighbouring atoms:
Dipole-Dipole Forces
-
Exist between polar molecules
Stronger than London dispersion forces
An attraction between the negative dipole of one molecule to the positive dipole of
another molecule
The force of attraction increases significantly between molecules as the distance
decreases
Hydrogen Bonding
-
Hydrogen bonding is a special case of dipole-dipole attractions
Occurs between molecules in which hydrogen is covalently bonded to a small, very
electronegative atom, principally nitrogen, oxygen or fluorine
Ion-Dipole Forces
-
An ion is attracted to an oppositely charged dipole of a polar molecule
The size and charge of the ion, as well as the magnitude of the dipole moment of the
polar molecule are factors in the strength of the ion-dipole force
Bonding and Molecular Shape
We’ll do these examples together so that everything makes sense!
Example 1: Chemical bonds are indicated below for some gaseous substances. Indicate which
atom will be more positive and which atom will be more negative in each bond. If both atoms
have equal positive or negative character, state that the bond is non-polar.
Na – Cl
C–F
P–O
C–C
K–H
H H
I I
H–C–C–H
I I
H H
Example 2: Arrange the following molecules in decreasing order of polarity.
F–F
C–F
H–F
C–H
Na – F
Example 3: Classify the bonds in each of the following substances as covalent, polar covalent, or
ionic:
K2 O
KCl
BeO
CBr4
KH
N2
SiF4
Lewis Structures, Molecular Shape, and Molecular Dipoles
1. For each of the following molecules:
i) Draw the Lewis structure and the molecular shape.
ii) Find ∆EN.
iii) Indicate the direction of any bond dipoles with vectors (arrows).
iv) Add the dipoles and determine if a molecular dipole exists.
a) LiF
b) CH4
c) HCl
d) Cl2
e) NH3
f) N2F4
g) BCl3
h) BeH2
i) H2Se
j) BF2H
h) SFCl5
i) CH2F2
2. Which of the following molecules has a molecular dipole?
a) NaF (g)
b) MgO (g)
c) MgI2 (g)
e) CO2 (g)
f) H2S (g)
g) CCl4 (g)
d) BCl3 (g)
h) PF3 (g)
 0
0



![QUIZ 2: Week of 09.03.12 Name: [7pts] 1.) Thoughtful list of 3](http://s3.studylib.net/store/data/006619037_1-3340fd6e4f1f4575c6d8cf5f79f0ff3e-300x300.png)