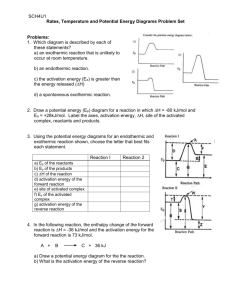
13.37 (1) The diagram in (a) shows the plots of ln k versus
1/T for two first-order reactions, where k is the rate
constant and T is the absolute temperature. Which
reaction has a greater activation energy? (2) The diagram
in (b) shows the plots for a first-order reaction at two
different temperatures. Which plot corresponds to a
higher temperature?
13.38 Given the same reactant concentrations, the
reaction
at 250°C is 1.50 × 103 times as fast as the same reaction
at 150°C. Calculate the activation energy for this
reaction. Assume that the frequency factor is constant.
13.40 Variation of the rate constant with temperature for
the first-order reaction
is given in the following table. Determine graphically the
activation energy for the reaction.
13.41 For the reaction
the frequency factor A is 8.7 × 1012 s−1 and the activation
energy is 63 kJ/mol. What is the rate constant for the
reaction at 75°C?
13.42 The rate constant of a first-order reaction is 4.60 ×
10−4 s−1 at 350°C. If the activation energy is 104 kJ/mol,
calculate the temperature at which its rate constant is
8.80 × 10−4 s−1.
13.43 The rate constants of some reactions double with
every 10-degree rise in temperature. Assume that a
reaction takes place at 295 K and 305 K. What must the
activation energy be for the rate constant to double as
described?
13.44 Consider the first-order reaction
Given that the frequency factor and activation energy for
the reaction are 3.98 × 1013 s−1 and 161 kJ/mol,
respectively, calculate the rate constant at 600°C.
13.45 Consider the second-order reaction
Given that the frequency factor and activation energy for
the reaction are 4.0 × 109/M · s and 85 kJ/mol,
respectively, calculate the rate constant at 500°C.
13.46 The rate at which tree crickets chirp is 2.0 × 102 per
minute at 27°C but only 39.6 per minute at 5°C. From
these data, calculate the “activation energy” for the
chirping process. (Hint: The ratio of rates is equal to the
ratio of rate constants.)
 0
0