Polar and Nonpolar Covalent Bonds
advertisement

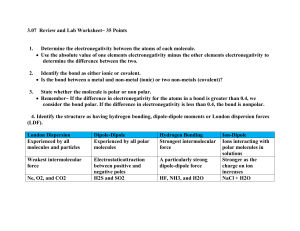
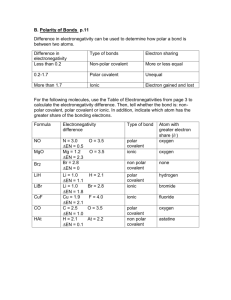
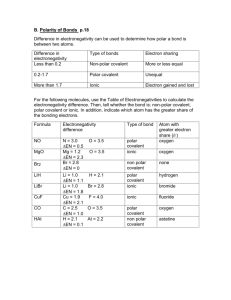
Polar and Nonpolar Covalent Bonds: The electrons in a covalent bond can be shared equally or unequally. The electronegativity values of the elements involved can tell you whether or not the electrons will be shared equally. Review: Define electronegativity – What is the trend in electronegativity as you go down a group on the periodic table? Across a period? Arrange the following elements in order of increasing electronegativity: a) Rb, Na, Li b) P, S, Al c) Fe, As, Br Nonpolar Covalent Bonds Both elements have the same electronegativity. They attract electrons with equal force, so the electrons are shared equally. Diatomic molecules are held together by nonpolar covalent bonds (Br2, O2 ,Cl2, I2 , F2, N2, H2 ) Write out the electron Dot Structures for the 7 Diatomic Elements. Bromine Iodine Oxygen Chlorine Nitrogen Fluorine Polar Covalent Bonds Each element has a different electronegativity value. (the sharing of electrons is unequal) Are polar because the element with the higher electronegativity will attract the shared electron more. The element that attracts the electron more will have a partially negative charge (it’s gaining an electron) The other element will appear to have a positive charge. Example: Hydrochloric Acid (HCl) Draw the dot diagram and decide which element is the more electronegative one (by using its placement on the periodic table.) For each of the following pairs of elements circle the one which is more electronegative: C and Cl F and N O and S H and Br Would the electronegativity difference between ionically-bonded elements be greater or less than covalently-bonded molecules? Why? The larger the electronegativity difference, the more polar and the more ionic character a bond is said to have. Remember, ionic bonds are also polar – they are the extreme in polarity because they don’t share at all. Nonpolar Polar Covalent Ionic Increasing Ionic Character and Polarity 0 difference 4.0 difference In each of the following, circle the bond with the most ionic character: a) C-Cl Na-Cl Cl-Cl c) C-F N-O Si-F b) N-F O-F d) H-Cl B-Cl S-Cl C-F In which compound does the bond between the atoms have the least ionic character? 1) HF 2) HCl 3) HBr 4) HI In which compound is the bond between the atoms the most polar? 1) HCl 2) NaCl 3) CCl4 4) AlCl3 Describe each of the following as either ionic or covalent and either polar or nonpolar: a) Mg-O c) O-O b) C-O d) N-O Draw the following molecules and label each bond as polar or nonpolar (p or np). If there is more than one polar bond, label which one has the most ionic character. HCCH Review – Draw LiCl SiH3Br