1.19 Electronegativity and Polarity _extra

SCH 4C
Remember :
Electronegativity and Polarity
A compound is a chemical combination of 2 or more elements. Compounds form to achieve stability through full outer energy levels (valence). Elements will move electrons to achieve this stability.
Electronegativity
Electronegativity (EN):
Circle the correct response.
Atoms have (different / identical) electronegativities. Electrons will spend more time near the atom with the (highest / lowest) electronegativity. The strongest electronegative element is (sodium / fluorine / rubidium). As you move across a (group / period) the electronegativity trend (increases / decreases / stays the same). As you move down a (group / period) the electronegativity trend (increases / decreases
/ stays the same). In general (metals / non metals) have lower electronegativities than (metals / non metals).
Remember that ionic bonds (transfer / share) electrons, while covalent bonds (transfer / share) electrons.
All atom’s (do / do not) attract electrons in the bond to the same degree. One can determine the degree to which electrons are transferred or shared using (electronegativity / electron affinity) differences. Each atom’s (electronegativity / electron affinity) has been determined through experimentation and can be found on (The Periodic Table / valence electrons). The difference is symbolized by (
EA /
EN). To find
(
EA /
EN) simply (add / subtract) the given values. If the (
EA /
EN) is between 3.3 and 1.7, the bond is considered (ionic / covalent / polar covalent).
When electrons are shared equally, the bond is considered to be (non polar / polar). This occurs when the (
EA /
EN) is between 0.00 and 0.5. An example of this type of bonding is (Oxygen gas / water).
When electrons are shared unequally, the bond is considered (non polar / polar) covalent. This is defined as (
EA /
EN) between 0.6 and 1.6. When the electrons are shared (equally / unequally), the result is a (partial / full) charge on the (more / less) electronegative element, as electrons spend more
‘time’ with that atom. This charge can be indicated using
. The more electronegative element would assume a slight (negative / positive) charge, while the least electronegative element would assume a slight (negative / positive) charge.
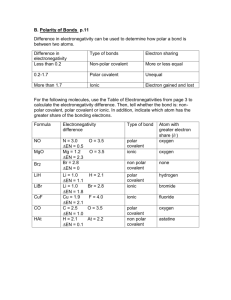
Identify the following compounds as Ionic, Polar Covalent or Non Polar Covalent.
A) B & P B) S & O C) Li & O D) Mg & N
Answers: A)0.15 non polar B) 0.86 polar C) 2.46 ionic D) 1.73 ionic




![QUIZ 2: Week of 09.03.12 Name: [7pts] 1.) Thoughtful list of 3](http://s3.studylib.net/store/data/006619037_1-3340fd6e4f1f4575c6d8cf5f79f0ff3e-300x300.png)