The Study of Matter Study Guide NAME: Physical Science 1
advertisement

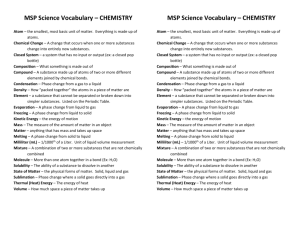
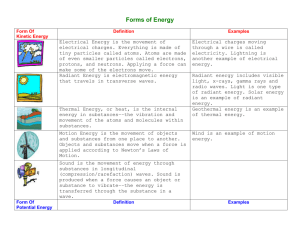
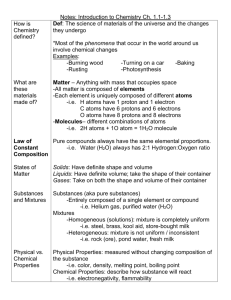
The Study of Matter Study Guide Physical Science NAME: ___________________________________ ____________________ 1. Everything in the universe is made of this ____________________ 2. This is the study of the composition of matter _____________________ 3. Give an example of something that does NOT have mass _____________________ 4. Anything that occupies space and has mass _____________________ 5. Is the amount of matter an object contains _____________________ 6. Are the simplest forms of matter _____________________ 7. This summarizes information about the elements _____________________ 8. Is the amount of force pulling down on an object _____________________ 9. Is the simplest particle of an element that retains all the properties of that element _____________________10. Each element has a different one _____________________11. Elements are made up of _______. _____________________12. This is located in the middle of an atom _____________________13. Name the two subatomic particles that make up the nucleus _____________________ _____________________14. This is the number of protons in an atom _____________________15. Particles with a positive charge _____________________16. This is the total number of protons and neutrons within an atom _____________________17. Particles with no charge _____________________18. Is a substance made of two or more different elements that are chemically bonded _____________________19. Particles with a negative charge _____________________20. Is the simplest part of a substance that retains all the properties of the substance and can exist in a free site _____________________21. These particles are very high in energy and have very little mass _____________________22. Shows the kinds and proportions of atoms of each element in a compound _____________________23. Where electrons are found The Study of Matter Study Guide NAME: ___________________________________ Physical Science _____________________24. Are molecules composed of atoms of the same element _____________________25. Is matter that has a fixed composition and definite properties _____________________26. The two types of pure substances _____________________ _____________________27. Are molecules composed of atoms of the same element _____________________28. The seven diatomic molecules _____________________ _____________________ _____________________ _____________________ _____________________ _____________________ _____________________29. Substances in which components are not distributed evenly _____________________30. Properties that you can observe or measure _____________________31. Properties that describe how a substance will react or change into a new substance _____________________32. A combination of two or more substances that are not chemically combined _____________________33. Substances in which the components are evenly distributed _____________________34. Is the temperature and pressure at which a solid becomes a liquid _____________________35. A homogeneous mixture of liquids _____________________36. Four examples of physical properties _____________________ _____________________ _____________________ _____________________37. A heterogeneous mixture of liquids The Study of Matter Study Guide NAME: ___________________________________ Physical Science _____________________38. Four examples of chemical properties _____________________ _____________________ _____________________ _____________________39. Solid, liquid or gas _____________________40. The units for density _____________________41. The temperature and pressure at which a liquid becomes a gas _____________________42. The formula for density _____________________43. Is the measure of the amount of matter in a certain volume of space _____________________44. When oxygen reacts with some metals forming an oxide _____________________45. Ability of a substance to combine chemically with another substance _____________________46. Ability to burn _____________________47. Ability to explode _____________________48. Four phases of matter _____________________ _____________________ _____________________ _____________________49. Is anything that affects one or more physical properties of a substance without changing its identity _____________________50. List the three ways we can separate mixtures, according to your book _____________________ _____________________ _____________________51. Occurs when 1 or more substances are completely changed into new substances _____________________52. During physical changes, this is released or absorbed The Study of Matter Study Guide NAME: ___________________________________ Physical Science _____________________53. List four examples of a physical change _____________________ _____________________ _____________________ _____________________54. List four examples of chemical changes _____________________ _____________________ _____________________ _____________________55. The three main branches of science _____________________ _____________________ _____________________56. A system of applied science _____________________57. A possible explanation based on multiple observations, experiments & reasoning _____________________58. The summary of many experimental observations and results _____________________59. Describe something using your senses _____________________60. Describe something using measurements or math equations _____________________61. A pattern, plan, representation, or description designed to show the structure or workings of an object, system or concept _____________________62. List three types of models _____________________ _____________________ _____________________63. Five things that are NOT matter _____________________ _____________________ _____________________ _____________________ The Study of Matter Study Guide Physical Science NAME: ___________________________________ _____________________64. Number of elements with official names _____________________65. The average mass of the atoms in an element Determine the number of atoms for each element in the following compounds. 66. C8H1608 67. NaOH 68. K3PO4 Solve each problem showing your work and labeling your answers. 69. A piece of titanium metal has a mass of 67.5 g and a volume of 15 cm3. What is the density of titanium? 70. A sample of a substance with a mass of 85 g occupies a volume of 110 cm3. What is the density of the substance? Will the substance float in water? Explain your answer. The Study of Matter Study Guide NAME: ___________________________________ Physical Science 71. Find the number of protons, electrons & neutrons for the following elements. A. Sulfur B. Tungsten C. Uranium D. Lithium Short Answer: 72. How does science take place? The Study of Matter Study Guide Physical Science 73. Draw a model of an atom. NAME: ___________________________________ 74. Compare and contrast physical changes and chemical changes.