NAME:________________________________
DATE:________________
PERIOD:_____
#: _____
Periodic Trends Notes & Activity
Valence Electrons
Valence electrons are ____________________________
______________________________________________
______________________________________________.
Group #
1
2
13
14
# of Valence Electrons
Group #
15
16
17
18
# of Valence Electrons
NOTE: Atoms / ions are most stable when they have __________ valence electrons.
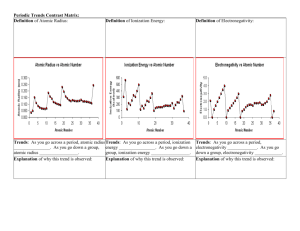
Atomic Radius
Atomic radius is _______________________________
_____________________________________________
_____________________________________________
_____________________________________________.
Graph the atomic radii of the elements from Group 1: Li = 1.52Å, Na = 1.54Å, K = 2.27Å, Rb =
2.48Å, and Cs = 2.65Å. Properly title graph and axes (with units). Note: Å = Angstrom.
2.5
2.0
1.5
1.0
Li
Na
K
Rb
Cs
Periodic Trends Notes & Activity
Q: What is the trend in atomic radius as you move down a group?
A: ____________________________________________________________________________
Q: Why does this trend occur?
Prediction
Actual Reason
Graph the atomic radii of the elements from Period 3: Na = 1.54Å, Mg = 1.36Å, Al = 1.25Å, Si =
1.17Å, P = 1.10Å, S = 1.04Å, and Cl = 0.99Å. Properly title graph and axes (with units).
2.0
1.5
1.0
0.5
Na
Mg
Al
Si
P
S
Cl
Q: What is the trend in atomic radius as you move right across a period?
A: ____________________________________________________________________________
Q: Why does this trend occur?
Prediction
Actual Reason
Periodic Trends Notes & Activity
Ionization Energy
Ions are atoms that have a positive or negative charge due to loss or gain of electrons.
Ionization energy is ______________________________________________________________
_____________________________________________________________________________.
A + Energy → _________________
Graph the ionization energies of the elements from Group 2: Be = 215, Mg = 176, Ca = 141, Sr =
131, Ba = 120, and Ra = 122. Properly title graph and axes (with units). Notes: units are
kcal/mol.
250
200
150
100
Be
Mg
Ca
Sr
Ba
Ra
Q: What is the trend in ionization energy as you move down a group?
A: ____________________________________________________________________________
Q: Why does this trend occur?
Prediction
Actual Reason
Periodic Trends Notes & Activity
Graph the ionization energies of the elements from Period 2: Li = 125, Be = 215, B = 191, C=
260, N= 336, O= 314, and F= 402. Properly title graph and axes (with units).
400
300
200
100
Li
Be
B
C
N
O
F
Q: What is the general trend in ionization energy as you move right across a period?
A: ____________________________________________________________________________
Q: Why does this trend occur?
Prediction
Actual Reason
Periodic Trends Notes & Activity
Electronegativity
Electronegativity is ______________________________________________________________
______________________________________________________________________________
Graph the electronegativities of the elements from Group 7: F = 4.0, Cl = 3.2, Br = 2.9,
and At = 2.2. Properly title graph and axes. Note: electronegativity has no units.
I = 2.7,
4
3
2
1
F
Cl
Br
I
At
Q: What is the trend in electronegativity as you move down a group?
A: ____________________________________________________________________________
Q: Why does this trend occur?
Prediction
Actual Reason
Periodic Trends Notes & Activity
Graph the electronegativities of the elements from Period 2: Li = 1.0, Be = 1.5, B = 2.0,
2.6, N = 3.1, O = 3.5 and F = 4.0. Properly title graph and axes.
C=
4
3
2
1
Li
Be
B
C
N
O
F
Q: What is the trend in electronegativity as you move right across a period?
A: ____________________________________________________________________________
Q: Why does this trend occur?
Prediction
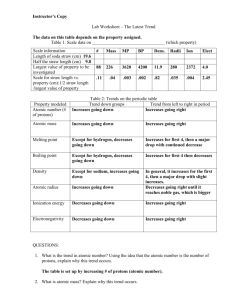
Trends Summary
Actual Reason
Periodic Trends Notes & Activity
Application Problems
1. What would have a greater atomic radius, phosphorus or bismuth?
2. What would have a smaller atomic radius, silver or strontium?
3. What would have a greater atomic radius, tin or chlorine?
4. Arrange the following elements from largest to smallest atomic radii: Li, O, C, K, Cs and F.
5. What would have a smaller ionization energy, titanium or zinc?
6. What would have a greater ionization energy, magnesium or barium?
7. What would have a greater ionization energy, sodium or rubidium?
8. Arrange the following elements from smallest to largest ionization energy: Li, O, C, K, Cs and F.
9. What would have a smaller electronegativity, carbon or lead?
10. What would have a greater electronegativity, tellurium or palladium?
11. What would have a smaller electronegativity, chlorine or mercury?
12. Arrange the following elements from smallest to largest electronegativity: Li, O, C, K, Cs and F.
 0
0