Experiment 20
Sodium Borohydride Reduction of a Ketone to an Alcohol
O
OH
1. NaBH4
2. H3O+
3,3-dimethyl-2-butanol
(Pinacoyl Alcohol)
3,3-dimethyl-2-butanone
(Pinacolone)
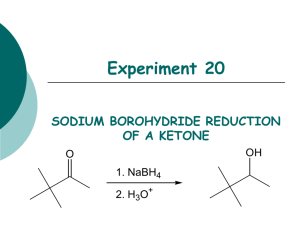
Figure 20.1 Reduction of a ketone to a secondary alcohol.
Introduction:
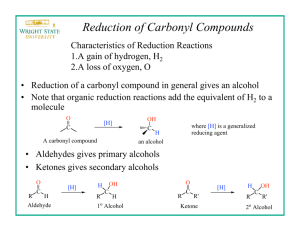
Reduction of a ketone using a metal hydride is an important reaction in organic chemistry
and one that is rapidly and easily performed, yet it is often ignored in organic lab manuals
(Figure 20.1). In today’s experiment, you will synthesize a secondary alcohol by reducing 3,3dimethyl-2-butanone with sodium borohydride for use in other experiments. Once synthesized
you will use a fractional distillation technique to purify the alcohol from unreacted starting
materials, and analyze the purity of your compound using GC analysis. NMR and IR spectra of
reactants and products are provided to aid in characterization of your compounds.
The most common laboratory reagents for the reduction of a carbonyl group of an
aldehyde or ketone to an alcohol are sodium borohydride (NaBH4) and lithium aluminum
hydride (LAH), shown in Figure 20.2. Both of these compounds behave as sources of a hydride
ion, which is a very strong nucleophile (see McMurry text, pages 609 and 709).
H
H
Na+H
B
H
Li+ H
Al
H
H
H
H
Sodium
borohydride
Lithium
aluminum
hydride
Hydride
ion
Figure 20.2 Common reducing agents.
Lithium aluminum hydride is a very powerful reducing agent and reacts not only with
aldehydes and ketones, but also with many other carbonyl containing compounds such as esters,
carboxylic acids, and amides. Another disadvantage of LAH is that it reacts violently with protic
solvents such as water and methanol, to produce metal hydroxides or alkoxides, and hydrogen
gas, which could result in an explosion or fire.
Sodium borohydride is much milder, much more selective reducing agent than lithium
aluminum hydride. Sodium borohydride will not reduce carbonyl-containing compounds that are
less reactive than aldehydes and ketones, such as esters, carboxylic acids, or amides, therefore
the aldehyde or ketone can be reduced with sodium borohydride in the presence of an acid or an
ester. These reductions can be performed in a wide variety of solvents, such as aqueous methanol
or ethanol, with a good to excellent yield.
169
The key step in the metal hydride reduction of an aldehyde or ketone is transfer of a
hydride ion from the boron atom of the reducing agent to the electropositive carbon of the
carbonyl group to form a tetrahedral intermediate (Figure 20.3). One mole of NaBH4 can react
with 4 carbonyls to give a tetraalkoxyborate. After the intermediate is hydrolyzed with water,
the tetraalkoxyborate is converted to the alcohol and boric acid salts. Thus, one mole of the
reducing agent reduces four moles of the carbonyl compound.
O
4 H 3C
H 3C
O
C
C
CH3
CH3
H
(from NaBH4)
H 3C
H 3C
H3O+
C
C
CH3
H
CH3
OH
4 H 3C
C
C
H 3C
CH
H 3
CH3
+
B(OH)3
Figure 20.3 Mechanism for the sodium borohydride reduction of a ketone.
IR Spectroscopy
In the IR spectra of alcohols, both the position of the OH stretch absorption and its
intensity depend on the extent of hydrogen bonding between alcohol molecules. Normally, this
hydrogen bonding is extensive, and the OH stretch appears as a broad peak at ~3200-3400 cm-1.
The C-O stretch absorption appears in the range of 1000-1300 cm-1. Also of importance are the
sp3 hybridized C-H stretch absorptions observed around 2850-3000 cm-1.
Ketones show a characteristic strong IR absorption between 1705-1725 cm-1 associated
with the C=O stretching absorption. Absorptions due to the sp3 hybridized C-H stretches are
also present. Use the provided IR spectra (Figure 20.4 and 20.5) to characterize the ketone and
the alcohol.
NMR Spectroscopy
The NMR spectra of the reactant and products are shown in Figure 20.4 and 20.5. The
chemical shift of a hydroxyl hydrogen in proton NMR varies depending on the purity of the
sample, the temperature, and the sample solvent. It typically appears in the range of 2.0-6.0
ppm, depending on the experimental conditions. Hydrogens on the carbon bearing the hydroxyl
group are deshielded by the oxygen atom, and typically appear in the range of 3.5-4.5 ppm.
Hydrogens attached to carbons to the carbonyl typically resonate between 2-3 ppm. One
distinguishing characteristic of a methyl ketone is the presence of a large, sharp three-proton
singlet around 2.1 ppm.
Objectives:
In this lab you will synthesize 3,3-dimethyl-2-butanol by a NaBH4 reduction of a ketone.
This product will be purified using fractional distillation. The identity and purity of the product
will be determined using GC analysis. Finally, you will use IR and NMR spectroscopy to
distinguish between reactants and products.
170
O
1.1
9H, singlet
4
2
3
1
4
4
2.1
2.1
3H,
singlet
3H, singlet
27
CHCl3
214
(NMR solvent)
Figure 20.4 1H, 13C, and IR spectra of 3,3-dimethyl-2-butanone.
171
45
24
0.9
9H, singlet
OH
4
2
3
1
4
1.1
3H, doublet
4
3.4
1H, quartet
2.2
1H, singlet
25
18
75
35
Figure 20.4 IR spectrum of 3,3-dimethyl-2-butanol.
1098
3400
2963
Figure 20.5 1H, 13C, and IR spectra of 3,3-dimethyl-2-butanol.
172
Experimental:
Synthesis:
Mix 2.0 mL of 3,3-dimethyl-2-butanone (pinacolone) and 10 mL of methanol in a 100 mL
beaker.
Place beaker in an ice water bath prepared in a 250 mL beaker.
Add 0.60 g of NaBH4 slowly while stirring with glass rod. Allow reaction to proceed for 5
minutes.
Remove beaker from ice bath and allow reaction to continue for an additional 10 minutes.
SLOWLY add 2.0 mL of 6M HCl dropwise to quench the excess NaBH4. A white precipitate
of boric acid should form (HCl will react with residual NaBH4 to produce H2 gas. It is
important to add HCl a few drops at a time to prevent bubbling over!)
Set up a suction filtration apparatus. Seat the filter paper with methanol. Slowly pour the
solution into the center of the Buchner funnel to remove the solid boric acid. If a significant
amount of solid appears in the liquid filtrate, filter again. If not, proceed to the next step.
Using a disposable pipette, transfer the liquid filtrate to a clean 50 mL round bottom flask
containing 2-3 boiling chips.
Purification and Product Isolation:
Prepare a fractional distillation apparatus (Appendix A). Set the VR @ 50, and collect
distillate that boils below 70oC into a small flask. This distillate is mainly methanol.
Switch receiving flasks to a preweighed 25 mL round bottom flask. Collect distillate that
boils between 70-85oC into this flask. This fraction should contain mainly the desired
product. Be sure to record the distillation range. DO NOT DISTILL TO DRYNESS!
After cooling, obtain a final product weight and calculate a final % yield. Complete Table
20.1 on the final lab report.
Product Analysis:
GC Analysis:
Prepare and submit a sample of your product for chromatographic analysis by placing
5 drops of your sample in an autoanalyzer vial and 1 mL of GC solvent (methanol).
Using chromatographic results propose an identity and determine the degree of purity
of your product. Complete Table 20.5 on the final lab report.
IR Analysis:
Using the provided, identify all characteristic absorptions of reactants and products.
Complete Table 20.3 on the final lab report.
NMR Analysis:
Using the provided spectra, identify and tabulate all characteristic resonances of
reactants and products. Complete Table 20.4 on the final lab report.
WASTE MANAGEMENT
Place the solid boric acid waste from the filtration into the container labeled “SOLID
WASTE” located in the waste hood. Place all liquid waste into the container labeled “LIQUID
WASTE”.
173
Final Lab Report
Due Date_______
Names______________________________
______________________________
Exp. 20: Sodium Borohydride Reduction of a Ketone
EXPERIMENTAL RESULTS
(Tables in INK only!)
Table 20.1 Experimental Results
Table 20.2 Green chemistry results
Theoretical yield (g)
Actual yield (g)
Percent yield
Distillation Range (oC)
Product Appearance
Atom Economy (%)
Experimental Atom
Economy (%)
“Eproduct”
Cost per synthesis ($)
Cost per gram ($/g)
Table 20.3 IR Analysis
Base Values
3,3-dimethyl-2butanone
Frequency (cm-1)
Frequency (cm-1)
3200-3400
2850-3000
1000-1300
1700-1725
Functional Group
OH stretch
sp3 CH stretch
C-O stretch
C=O stretch
3,3-dimethyl-2butanol
Frequency (cm-1)
Table 20.4 NMR Analysis
3,3-dimethyl-2-butanone
3,3-dimethyl-2-butanol
C1
O
OH
H1
C2
2
3
H1
C1
2
C2
3
1
1
4
C3
4
H4
OH
4
4
4
H2
C3
4
C4
C4
H4
Table 20.5 GC Analysis
Compound
GC Retention times (min)
Standards
Sample
methanol
3,3-dimethyl-2-butanone
3,3-dimethyl-2-butanol
174
Area percent
Adjusted
Area Percent
DISCUSSION/CONCLUSIONS
(In the space provided, briefly answer the following questions. Use numerical values to support conclusions where applicable.)
O
NaBH4
Sodium borohydride
Acros # 20005
Cost: $112.20/ 100g
OH
CH3OH
methanol
Acros # 42395
Cost: $27.00/ 500mL
3,3-dimethyl-2-butanone
Acros # 13125
Cost: $21.20/ 100mL
6M HCl
hydrochloric acid
Acros # 42379
Cost: $27.80/ 500mL
3,3-dimethyl-2-butanol
Acros # 15968
Cost: $13.10/ 10g
1.
Based on GC analysis, did your sample contain pure product? Explain using actual adjusted area percent
values.
2.
What type of absorption band would be present in the IR spectrum of the product if unreacted 3,3-dimethyl2-butanone was present? What is the typical frequency for this type of absorption? What is the actual
frequency of the absorption in the provided spectrum?
3.
What is one signal in the 1H NMR spectrum that indicates that the reduction took place? What are the
typical chemical shifts that these types of protons may appear? What is the actual chemical shift of this
proton signal?
4.
Draw the product, and a complete mechanism for its formation, for the following reaction:
O
H
CH3
Na
H3C
C CH2 CHCH2CH3
H
B
H
H
**Attach sample chromatogram!**
175
176
 0
0