Periodic Trends Lab
advertisement

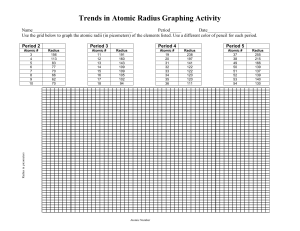
Name: Period: 7 Date: Periodic Trends: A Graphical Analysis Introduction Elements on the periodic table are arranged in such a way that they exhibit patterns in their properties. In this activity, you will graph 3 periodic properties and analyze the patterns. In this activity, you will graph 3 periodic properties and analyze the patterns. You will determine trends for the following properties: atomic radius, electronegativity, and ionization energy. Definitions Go to the online textbook. Click on the word “Glossary” at the top. Use the glossary to define the following terms. 1. Electronegativity: 2. Atomic Radius: 3. Ionization Energy: 4. Period: 5. Group: Graphing #1: Electronegativity 1. Open a spreadsheet in Excel. 2. In column A, going down, type the numbers 1-18. These numbers represent the atomic numbers for the first 18 elements on the periodic table. 3. In column B, going down, type the following set of numbers (in order!): 2.2, 0, 1, 1.5, 2, 2.5, 3, 3.5, 4, 0, 0.9, 1.2, 1.5, 1.8, 2.1, 2.5, 3, 0. These numbers represent the electronegativity values for the first 18 elements. 4. Highlight all of the data in columns A and B. 5. Click on the “Chart Wizard” icon on the toolbar (it looks like a bar graph). If this icon is not on your toolbar, you can access the chart wizard using the menu by choosing Insert…Chart. 6. Under Chart Type, choose XY (Scatter). Under Chart Sub-type, choose the picture on the left-hand side of the bottom row. Click Next. 7. Click Next. 8. Type “Electronegativty” for Chart title, “Atomic Number” for Value (X) axis, and “Electronegativity” for Value (Y) axis. Click Finish. Electronegativity Analysis Questions 1. Examine your graph. Find the atomic numbers #3-9 that correspond to the elements in period 2. What happens to the electronegativity values as you move across the period? 2. Examine your graph. Find the atomic numbers #11-17 that correspond to the elements in period 3. What happens to electronegativity values as you move across the period? 3. Examine your graph. Find the atomic numbers #1, #3, and #11 that correspond to the elements in Group 1 (Hydrogen, Lithium, and Sodium). What happens to electronegativity as you move down the group? 4. Summary of trends for electronegativity: Electronegativity and down a group. across a period Graphing #2: Atomic Radius 1. Return to your spreadsheet. At the bottom, click on the tab that says Sheet2. 2. In column A, going down, type the numbers 1-18. These numbers represent the atomic numbers for the first 18 elements on the periodic table. 3. In column B, going down, type the following set of numbers (in order!): 0.3, 0.93, 1.52, 1.11, 0.88, 0.77, 0.7, 0.66, 0.64, 1.12, 1.86, 1.6, 1.43, 1.17, 1.1, 1.04, 0.99, 1.54. These numbers represent the atomic radius values ( in x 10-10 m) for the first 18 elements. 4. Highlight all of the data in columns A and B. 5. Click on the “Chart Wizard” icon on the toolbar (it looks like a bar graph). If this icon is not on your toolbar, you can access the chart wizard using the menu by choosing Insert…Chart. 6. Under Chart Type, choose XY (Scatter). Under Chart Sub-type, choose the picture on the left-hand side of the bottom row. Click Next. 7. Click Next. 8. Type “Atomic Radius” for Chart title, “Atomic Number” for Value (X) axis, and “Atomic Radius” for Value (Y) axis. Click Finish. Atomic Radius Analysis Questions 1. Examine your graph. Find the atomic numbers #3-10 that correspond to the elements in period 2. What happens to atomic radius values as you move across the period? 2. Examine your graph. Find the atomic numbers #11-18 that correspond to the elements in period 3. What happens to atomic radius values as you move across the period? 3. Examine your graph. Find atomic numbers #1, #3, and #11 that correspond to the elements in Group 1 (Hydrogen, Lithium, and Sodium). What happens to atomic radius as you move down the group? 4. Summary of trends for atomic radius: Atomic Radius and down a group. across a period Graphing #3: Ionization Energy 1. Return to your spreadsheet. At the bottom, click on the tab that says Sheet3. 2. In column A, going down, type the numbers 1-18. These numbers represent the atomic numbers for the first 18 elements on the periodic table. 3. In column B, going down, type the following set of numbers (in order!): 314, 567, 124, 214, 191, 261, 334, 314, 401, 498, 118, 175, 138, 187, 254, 240, 300, 364. These numbers represent the ionization energy values in kcal/mol for the first 18 elements. 4. Highlight all of the data in columns A and B. 5. Click on the “Chart Wizard” icon on the toolbar (it looks like a bar graph). If this icon is not on your toolbar, you can access the chart wizard using the menu by choosing Insert…Chart. 6. Under Chart Type, choose XY (Scatter). Under Chart Sub-type, choose the picture on the left-hand side of the bottom row. Click Next. 7. Click Next. 8. Type “Ionization Energy” for Chart title, “Atomic Number” for Value (X) axis, and “Ionization Energy” for Value (Y) axis. Click Finish. Ionization Energy Analysis Questions 1. Examine your graph. Find the atomic numbers #3-10 that correspond to the elements in period 2. What happens to ionization energy values as you move across the period? 2. Examine your graph. Find the atomic numbers #11-18 that correspond to the elements in period 3. What happens to ionization energy values as you move across the period? 3. Examine your graph. Find atomic numbers #1, #3, and #11 that correspond to the elements in Group 1 (Hydrogen, Lithium, and Sodium). What happens to ionization energy as you move down the group? 4. Summary of trends for ionization energy: Ionization Energy a period and down a group. To Submit Your Work: Copy all 3 graphs into this Word document (you can just paste them in at the end). Save the document as LastNamePTLab.doc . across