Quantum Numbers Wkst

Chem 1A Handout: Chapter 7 Study Information
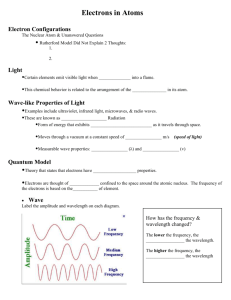
Quantum Numbers:
Each electron in an atom can be represented by a set of four quantum numbers.
These numbers compliment the information given as the electron configuration and set the limits as to how many electrons can be placed in one orbital as two; or the total number of electrons in any subshell (for example the d-subshell has 10 electrons because there are ten possible combinations of quantum numbers).
The ultimate rule which summarizes the experiences of electron configurations from quantum numbers is called the Pauli Exclusion Principle –stating that no two electrons can have the same four quantum numbers.
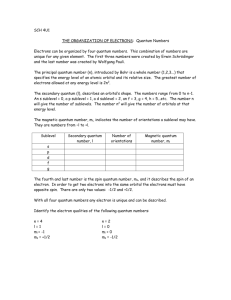
1. Principle Quantum number , n has values of any integer from 1 to
∞
(represents the energy level or shell)
2. Angular Momentum Quantum number , l has integral values from 0 to (n-1) l values on the 7th energy level: 0, 1, 2, 3, 4, 5, 6 subshell designations: s, p, d, f, g, h, i
3. Magnetic Quantum number , m l has integral vales from l to 0 to + l.
(describes the orientation of an orbital in space)
4. Electron spin quantum number , m s
only has values of +1/2 or - 1/2
(two possible spinning motions of an electron, spin up and spin down)
Questions:
1. For n=6, what are all the possible values of l.
(this represents all the possible energy sublevels on the 6th energy level or shell)
write out these designations as subshells:
2. For l = 4, what are the possible values of m l .
How many orbitals are in this sublevel?
3. Write out 14 sets of four quantum numbers which designate all the electrons known to be possible in the 4
f subshell?
n= l = m l = ms=
4. Decide if the following sets of quantum numbers are permissible, if not signify the wrong value or values
by circling it and then explaining what was wrong.
a. (2, 0, 0, +1/2 )
b. (3, -1, 0, +1/2 )
c. (7, 5, -4, -1/2 )
d. (2, 0, 0, -1/2 )
e. (9, 6, -7, +1/2 )
4b. Which of the above represent electrons in the same orbital and having the same energy?
Electron Configurations:
1. Write out the complete electron configuration for osmium, (Os). b) Now write out the shorthand notation, with the noble gas inner core abbreviation and the remaining
electron configuration. c) Draw out in orbital notation (as arrows) in labeled orbitals, all electrons that are not part
of the noble gas inner core. d) How many valence electrons are in the osmium atom. In which energy level do these occur? e) How many unpaired electrons are in the ground state electron configuration of osmium?
Is osmium diamagnetic or paramagnetic? Explain your answer.