Quantum number note

From Bohr to Schrodinger…
Bohr model had a number of successes (which is why we still use it so much!)
explained the line spectra of the H-atom
predicted accurately bonding between valence electrons for many elements
But there were also some problems and things it could not explain…
line spectra of more complex atoms
why some transition metals had multiple charges (Fe for example)
why there are some anomalies in the periodic trends (IE for example)
Schrodinger and others refined Bohr’s model to develop our modern quantum model of the atom (reread your handout for some of the details.). In this model, each electron is assigned 4 quantum numbers. These numbers are ‘variables’ in the Schrodinger wave equation and the result is an ‘area of probability space’ where that electron is found – called an orbital.
Miscellaneous...
Lots of cool stuff owes itself to quantum….
X-rays… Sort of like the opposite of the photoelectric effect.
In the photoelectric effect, light can cause electrons to be released from a material
In making an X-ray, electrons are shot at certain materials and X-ray light is released
More later – but check out: http://www.colorado.edu/physics/2000/index.pl
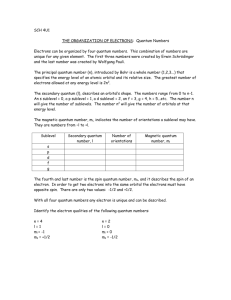
The quantum numbers:
1.
Principal quantum number – n. Goes from n=1 to n=∞
- this number describes the orbital (or shell) that the electron is found in.
2.
Orbital shape quantum number – l
. Goes from 0 to n-1
Value of l
0
- this number describes the subshell. Each subshell has a distinct shape, these are given letter names.
Letter designation s
1
2
3 p d f picture
3.
Magnetic quantum number – m l
. Goes from –
l
to +
l
- this describes the orientation of the subshell. The number of orientations depends on the value of l
. For s, there is only 1 orientation. For p there are 3. How many would there be for d, f and g?
4.
Spin quantum number – m s
. Is either +½ or -½
- after Shrodinger came more research (Heisenberg, Pauli.). Pauli’s work showed that no 2 electrons could have the same 4 quantum number address. This meant that each orbital can only hold 2 electrons.
So, for the single electron in a hydrogen atom it would have the 4 quantum numbers:
1, 0, 0, +½
What would the 3 rd electron in lithium be?
What would this describe?
2, 1,1, +½