Certain arrangements of valence electrons are
more stable than others.
The valence electron configurations of the noble
gases are considered the most stable of all
valence electron configurations.
1
Chemical Bonding
Atoms
gain, lose or share valence
electrons in order to obtain the same
number of electrons as a noble gas
A Chemical Bond
Chemical Bond - Attractive force that holds two
atoms together in a compound
Bonds form as a result of interactions between
valence electrons found in the combining atoms.
3
Introduction to Bonding
Consider
2 classes of bonds:
Ionic bonding
Covalent bonding
Ionic Bonds
bonds – attractive forces among
oppositely charged ions
Ionic
Forms when a metal loses electron(s) to a
nonmetal.
The bond is stronger for small ions and for
ions with larger charges
• NaF vs. MgO
Ion Review
Ion – a charged atom (or group of atoms) that is
electrically charged as a result of the loss or gain
of electrons.
If an atom gains one or more electrons, it becomes a
negatively charged ion.
If an atom loses one or more electrons, it becomes a
positively charged ion.
6
Metal atoms containing one, two, or three
valence electrons lose electrons to obtain a
noble-gas electron configuration.
Group
Charge
IA
1+
IIA
2+
IIIA
3+
7
Nonmetal atoms containing five, six, or seven
valence electrons gain electrons to obtain a
noble-gas electron configuration.
Group
Charge
VIIA
1–
VIA
2–
VA
3–
8
The Reaction Between Sodium
and Chlorine
The Reaction Between Sodium
and Oxygen
The Reaction Between Calcium
and Chlorine
Copyright © Cengage
Learning. All rights
reserved
11
Ionic Compounds
Consists of positive and negative ions arranged
in such a way that each ion is surrounded by
nearest neighbors of the opposite charge.
Any given ion is bonded to all the other ions of
opposite charge surrounding it.
Cross-Section of NaCl
Sodium Chloride (NaCl)
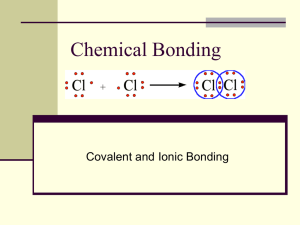
Covalent Bonds
bond – bonded atoms share
pairs of valence electrons
Covalent
Covalent bonding results in formation of a
molecule.
Covalent bonding occurs between nonmetals.
Covalent Bonds
bond – bonded atoms share 1 pair
of valence electrons
Single
Double
bond– bonded atoms share 2 pairs
of valence electrons
Triple
bond– bonded atoms share 3 pairs
of valence electrons
Describing Covalent Bonds
energy – energy needed to
separates covalently bonded atoms
Bond length – distance between the nuclei
of bonded atoms
Bond
The more electrons shared by a given pair of
atoms the stronger and shorter the bond
Types of Covalent Bonds
1. Nonpolar covalent bond – covalent bond in
which there is equal sharing of electrons
between bonded atoms.
2. Polar covalent bond – covalent bond in which
there is unequal sharing of electrons between
bonded atoms.
Bond Polarity
To
predict bond polarity - compare the
electronegativity (EN) of the bonded
atoms.
EN – the ability of an atom in a molecule to
attract shared electrons.
The higher the EN the better the atom attracts
shared electrons
Polar Covalent Bonds
Electrons spend more time near the more
electronegative atom of the bond and less
time near the less electronegative atom of the
bond.
Polar Covalent Bond
Unequal sharing of electrons creates partial
positive and negative charges on atoms.
Two ways to show bond polarity:
Consider the HCl molecule.
EN and Bond Polarity
The
greater the difference in EN between
bonded atoms the more polar the bond.
Bond polarity and bond type impacts the
physical properties of molecules.
More polar molecules have higher boiling
points
Ionic compunds have even higher boiling
points.
Bond Polarity
Bond Type
Electronegativity Difference
Nonpolar Covalent*
less than 0.5*
Polar Covalent
0.5 to 1.5
Polar Covalent
Between 1.5 and 2.0
(between nonmetals)
Ionic
Between 1.5 and 2.0
(metal and nonmetal)
Ionic
Greater than 2.0
*see next slide
*Polarity Comments
Zumdahl classifies bonds with very small EN
differences to be very slight polar – and he’s
correct…
HOWEVER, bonds with an EN difference of
less than 0.5 (and more than 0) behave in
essentially the same way as nonpolar bonds.
Therefore, many (me included) classify these
bonds to be nonpolar or essentially nonpolar.
Practice
Arrange the following bonds from most to
least polar:
C-H
C-F
N-O
Si-F
N-N
Label the polarity in each of the polar bonds
Which of the following bonds would be the
least polar yet still be considered polar
covalent?
Mg-O
C-O
O-O
Si-O
N-O
27
Which of the following bonds is the most
polar without being considered ionic?
Mg-O
C-O
O-O
Si-O
N-O
Key Differences Between Ionic
and Covalent Bonding
Ionic bonds form between a metal and
nonmetal. Covalent bonds usually form between
nonmetals.
2. Ionic bonds involve electron transfer. Covalent
bonds involve electron sharing.
3. Ionic compounds contain ions and not discrete
molecules. A covalent compound has a
molecule as its basic structural unit (and no
ions).
1.
Key Differences Between Ionic
and Covalent Bonding
All ionic compounds are solids at room
temperature. Covalent compounds are varied.
5. Soluble ionic solids form aqueous solutions that
conduct electricity. Soluble covalent compounds
usually produce a nonconducting aqueous
solution.
4.
Lewis Structures
Lewis
structures can be drawn for both
molecules and ions.
Lewis Structures show how the atoms in
molecules and polyatomic ions are:
Arranged
Bonded to each other
Lewis
Structures show:
All atoms in the molecule or ion
All valence electrons.
• For ions:
An electron must be added to the total # of valence e for
each negative charge
An electron must be subtracted from the total # of valence
electrons for each positive charge
An octet (8) electrons around each atom, but
hydrogen and a duet (2) electrons for
hydrogen.
Lewis Structures
See
board for examples of correctly (and
incorrectly) drawn Lewis structures.
Interparticle Forces
In
the liquid and solid state the attractive
forces between particles are strong
enough to hold the particles together.
Interparticle Forces and
Physical Properties
The
stronger the attractive forces between
particles in a liquid or solid, the
Higher the:
•
•
•
•
Melting point
Boiling point
Surface tension
Viscosity
Lower the:
• Vapor pressure
Ionic Compounds
For
ionic compounds the force between
particles is the ionic bond
Creates a 3-D network of STRONG attractive
forces between oppositely charged ions
As a result of these attractions the ions are
locked in place.
Ionic Compounds
Ionic
bonds are the strongest interparticle
force.
We find that the smaller the ion and the larger
the charge on the ion the stronger the ionic
bond
Molecules
Forces – strong attractive
forces within the molecule hold the atoms
in a fixed ratio
Intramolecular
Also called ______________
• Already studied these
(IM) Forces – relatively
weak attractive forces between individual
molecules
Intermolecular
Forces Between Particles
Ionic compounds
Attractive forces between ions hold ionic compounds
together.
Ionic bonds are the strongest interparticle force.
Molecular compounds
IM forces between molecules attract molecules to
each other in the liquid and solid state.
• IM forces are very weak as compared to ionic bonds
IM Forces
Why
are IM forces only of importance to
molecular compounds in the liquid or solid
state?
IM Forces
Three types of IM Forces
1.
2.
3.
Dipole-dipole force
Hydrogen “bonding”
Dispersion forces
IM Forces
Dipole-dipole
forces
Attractive forces between oppositely charged
dipoles.
Dipole-dipole forces are found between polar
compounds.
• The more polar the compound the stronger the
dipole-dipole force.
IM Forces
Hydrogen
bonds
Attractive force between a d+ H bonded to an
O, N, or F and a d- O, N, or F on another
molecule.
• Not a true bond….
Hydrogen Bonding
Hydrogen
bonding is a type of ________
force.
Hydrogen bonding is the strongest of the IM
forces.
H bonding is very important in water and in
biological molecules.
• FYI – large biological molecules often H bond to
themselves.
H Bonding
Water
Also called the water network
NH3
Solution
of alcohol and water
Dispersion Forces
Dispersion
force
Very weak and short-lasting attractive forces
between momentary and induced dipoles.
Weakest of the IM forces
London Dispersion Forces
Dispersion
force
Found between all molecules.
• Of greatest significance in nonpolar and large
atoms/molecules
• Only IM force between nonpolar molecules
The larger the molecule the stronger the
dispersion forces.
Dispersion Forces
Momentary and induced dipoles are not the
same as the “permanent” dipoles found in
polar bonds.
See board
London Dispersion Forces
A weak temporary intermolecular force.
Results from momentary uneven electron
distributions of electrons in molecules.
Occurs between all molecules.
Significant in large atoms/molecules and
nonpolar molecules.
Larger the atom/molecule the stronger the force.
Interparticle Forces
Weakest:
Strongest:
Interparticle Forces and
Physical Properties
The
stronger the attractive forces between
particles in a liquid or solid, the
Higher the:
•
•
•
•
Melting point
Boiling point
Surface tension
Viscosity
Lower the:
• Vapor pressure