File
advertisement

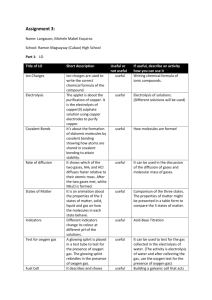
Patterns of Chemical Change Rates of Reaction Chemical reactions occur when different atoms or molecules collide: For the reaction to happen the particles must have a certain amount of energy – this is called the ACTIVATION ENERGY. The rate at which the reaction happens depends on four things: 1) The temperature of the reactants, 2) Their concentration 3) Their surface area 4) Whether or not a catalyst is used Measuring rate of reaction Two common ways: 1) Measure how fast the products are formed 2) Measure how fast the reactants are used up Rate of reaction graph Amount of product formed Fast rate of reaction here Slower rate of reaction here due to reactants being used up Slower reaction Time Biological catalysts Our bodies contain catalysts for all the reactions that go on inside them – they are called ENZYMES. Enzymes work best at certain temperatures: Optimum temperature Enzyme (roughly 40OC) activity Enzymes are denatured beyond 40OC Increasing activity due to more collisions Temperature Enzymes are used in the food industry. Two examples: 1) Fermentation: Glucose Ethanol + Carbon dioxide Ethanol is used for alcohol, carbon dioxide for making bread rise 2) Yoghurt production – enzymes in bacteria produce yoghurt from milk by converting sugar (lactose) into lactic acid, which tastes sour Endothermic and exothermic reactions Step 1: Energy must be SUPPLIED to break bonds: Step 2: Energy is RELEASED when new bonds are made: A reaction is EXOTHERMIC if more energy is RELEASED then SUPPLIED. If more energy is SUPPLIED then is RELEASED then the reaction is ENDOTHERMIC Energy level diagrams Energy level Activation energy Using a catalyst might lower the activation energy Energy given out by reaction Reaction progress Exothermic vs endothermic: EXOTHERMIC – more energy is given out than is taken in (e.g. burning, respiration) ENDOTHERMIC – energy is taken in but not necessarily given out (e.g. photosynthesis) Burning Methane CH4 + 2O2 To burn methane you have to break all of these bonds: And then you have to make these ones: 2H2O + CO2 Bond energies C-H = 435 Kj O=O = 497 Kj Total for breaking bonds = 4x435 + 2x497 = 2734 Kj C=O = 803 Kj H-O = 464 Kj Total for making bonds = 2x803 + 4x464 = 3462 Kj Drawing this on an energy diagram: 2734 Kj 3462 Kj -728 Kj More energy is given out (3462) than is given in (2734) – the reaction is EXOTHERMIC. The total (“nett”) energy change is –728 Kj. An endothermic reaction would have a positive energy change. Atomic mass RELATIVE ATOMIC MASS, Ar (“Mass number”) = number of protons + number of neutrons SYMBOL PROTON NUMBER = number of protons (obviously) Relative formula mass, Mr The relative formula mass of a compound is blatantly the relative atomic masses of all the elements in the compound added together. E.g. water H2O: Relative atomic mass of O = 16 Relative atomic mass of H = 1 Therefore Mr for water = 16 + (2x1) = 18 Work out Mr for the following compounds: 1) HCl H=1, Cl=35 so Mr = 36 2) NaOH Na=23, O=16, H=1 so Mr = 40 3) MgCl2 Mg=24, Cl=35 so Mr = 24+(2x35) = 94 4) H2SO4 H=1, S=32, O=16 so Mr = (2x1)+32+(4x16) = 98 5) K2CO3 K=39, C=12, O=16 so Mr = (2x39)+12+(3x16) = 138 More examples CaCO3 40 + 12 + 3x16 HNO3 1 + 14 + 3x16 2MgO 2 x (24 + 16) 3H2O 3 x ((2x1) + 16) 4NH3 2KMnO4 3C2H5OH 4Ca(OH)2 100 80 Calculating percentage mass If you can work out Mr then this bit is easy… Percentage mass (%) = Mass of element Ar Relative formula mass Mr x100% Calculate the percentage mass of magnesium in magnesium oxide, MgO: Ar for magnesium = 24 Ar for oxygen = 16 Mr for magnesium oxide = 24 + 16 = 40 Therefore percentage mass = 24/40 x 100% = 60% Calculate the percentage mass of the following: 1) Hydrogen in hydrochloric acid, HCl 2) Potassium in potassium chloride, KCl 3) Calcium in calcium chloride, CaCl2 4) Oxygen in water, H2O Calculating the mass of a product E.g. what mass of magnesium oxide is produced when 60g of magnesium is burned in air? Step 1: READ the equation: 2Mg + O2 2MgO IGNORE the oxygen in step 2 – the question doesn’t ask for it Step 2: WORK OUT the relative formula masses (Mr): 2Mg = 2 x 24 = 48 2MgO = 2 x (24+16) = 80 Step 3: LEARN and APPLY the following 2 points: 1) 48g of Mg makes 80g of MgO 2) 60g of Mg makes 60/48 x 80 = 100g of MgO 1) When water is electrolysed it breaks down into hydrogen and oxygen: 2H2O 2H2 + O2 What mass of hydrogen is produced by the electrolysis of 6g of water? 1. Work out Mr: 2H2O = 2 x ((2x1)+16) = 36 2H2 = 2x2 = 4 2. 36g of water produces 4g of hydrogen, 3. 6g of water will produce (6/36) x 4 = 0.66g of hydrogen 2) What mass of calcium oxide is produced when 10g of calcium burns? Mr: 2Ca = 2x40 = 80 2Ca + O2 2CaO 2CaO = 2 x (40+16) = 112 80g produces 112g so 10g produces (112/80) x 10 = 14g of CaO 3) What mass of aluminium is produced from 100g of aluminium oxide? 2Al2O3 Mr: 2Al2O3 = 2x((2x27)+(3x16)) = 204 4Al + 3O2 4Al = 4x27 = 108 204g produces 108g so 100g produces (108/204) x 100 = 52.9g of Al2O3 Another method Try using this equation: Mass of product IN GRAMMES Mass of reactant IN GRAMMES Mr of product Mr of reactant Q. When water is electrolysed it breaks down into hydrogen and oxygen: 2H2O 2H2 + O2 What mass of hydrogen is produced by the electrolysis of 6g of water? Mass of product IN GRAMMES 6g 4 36 So mass of product = (4/36) x 6g = 0.66g of hydrogen Calculating the volume of a product At normal temperature and pressure the Relative Formula Mass (Mr) of a gas will occupy a volume of 24 litres e.g. 2g of H2 has a volume of 24 litres 32g of O2 has a volume of 24 litres 44g of CO2 has a volume of 24 litres etc Q. When water is electrolysed it breaks down into hydrogen and oxygen: 2H2O 2H2 + O2 What VOLUME of hydrogen is produced by the electrolysis of 6g of water? • On the previous page we said that the MASS of hydrogen produced was 0.66g • 2g of hydrogen (H2) will occupy 24 litres (from the red box above), • So 0.66g will occupy 0.66/2 x 24 = 8 litres Example questions 1) What volume of hydrogen is produced when 18g of water is electrolysed? 2H20 2H2 + O2 2) Marble chips are made of calcium carbonate (CaCO3). What volume of carbon dioxide will be released when 500g of CaCO3 is reacted with dilute hydrochloric acid? CaCO3 + 2HCl CaCl2 + H2O + CO2 3) In your coursework you reacted magnesium with hydrochloric acid. What volume of hydrogen would be produced if you reacted 1g of magnesium with excess acid? Mg + 2HCl MgCl2 + H2 Empirical formulae Empirical formulae is simply a way of showing how many atoms are in a molecule (like a chemical formula). For example, CaO, CaCO3, H20 and KMnO4 are all empirical formulae. Here’s how to work them out using masses: A classic exam question: Find the simplest formula of 2.24g of iron reacting with 0.96g of oxygen. Step 1: Divide both masses by the relative atomic mass: For iron 2.24/56 = 0.04 For oxygen 0.96/16 = 0.06 Step 2: Write this as an equation and simplify: 0.04:0.06 is equivalent to 2:3 Step 3: Write the formula: 2 iron atoms for 3 oxygen atoms means the formula is Fe2O3 Example questions 1) Find the empirical formula of magnesium oxide which contains 24g of magnesium and 16g of oxygen. 2) Find the empirical formula of a compound that contains 42g of nitrogen and 9g of hydrogen. 3) Find the empirical formula of a compound containing 20g of calcium, 6g of carbon and 24g of oxygen. Electrolysis Molecule of solid copper chloride Molecule of solid copper chloride after being dissolved Chloride ion Copper ion Electrolysis Electrolysis is used to extract a HIGHLY REACTIVE metal. When we electrolysed copper chloride the negative chloride ions moved to the positive electrode and the positive copper ions moved to the negative electrode – OPPOSITES ATTRACT!!! = chloride ion = copper ion Electrolysis equations We need to be able to write “half equations” to show what happens during electrolysis (e.g. for copper chloride): At the negative electrode the positive ions GAIN electrons to become neutral copper ATOMS. The half equation is: Cu2+ + 2 e- Cu At the positive electrode the negative ions LOSE electrons to become neutral chlorine MOLECULES. The half equation is: 2 Cl- - 2 e- Cl2 Calculating masses and volumes from electrolysis Example question: How much chlorine is released at the positive electrode if 2g of copper is collected at the negative electrode? Consider those two half equations again: Cu2+ + 2e- Cu 2Cl- - 2e- Cl2 1) Write down the relative atomic mass: Copper = 63 2 molecules of chlorine (Cl2) = 2x35 = 70 2) Now use this equation: Actual mass of chlorine Relative mass of chlorine Actual mass of copper Relative mass of copper 3) Write down the answer: _______g of chlorine is liberated, which would occupy a volume of ________litres