Optical isomers
advertisement

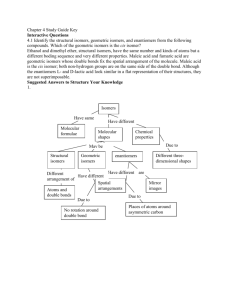
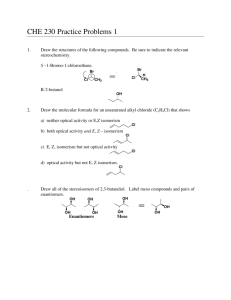
Topic 20 High Level Organic Chemistry IB HL Objective • 20.1.1 Deduce structural formulas for compounds containing up to six carbon atoms with one of the following functional groups: amine, amide, ester and nitrile. • 20.1.2 Apply IUPAC rules for naming compounds containing up to six carbon atoms with one of the following functional groups: amine, amide, ester and nitrile. Amines (R-NH2) Prefix: aminoOr for the first three carbons: Suffix: -ylamine H R N Examples: CH3CH2NH2 ethylamine CH3CH(NH2)CH2CH2CH3 2-aminopentane H Amide (-CONH2) Suffix: -anamide Example: CH3CONH2 ethanamide O R C NH2 Ester (-COOC-) Because esters are produced from alcohols heated with carboxylic acids… The chain from the alcohol provides first part of the name, and the chain from the carboxylic acid is given the name of the acid anion. Example: CH3-CO-O-CH3 methyl ethanoate Nitriles (R-CN) Used to be called cyanides. Suffix: -nitrile added to the hydrocarbon forming the basis of the acid. (Sometimes you will see the prefix cyano-) Example: CH3CH2CN propanenitrile CH3CN ethanenitrile IB HL Objective • 20.2.1 Explain why the hydroxide ion is a better nucleophile than water. 20.2.1 Explain why the hydroxide ion is a better nucleophile than water. • Nucleophilic: What’s in a name? • Nucleo—nucleus philic--loving • So nucleophiles are molecules which are attracted to more positive charge. • Nucleophiles will then donate electrons. 20.2.1 Explain why the hydroxide ion is a better nucleophile than water. Water vs. Hydroxide H2O and OH• When a carbon is attached to a more electronegative atom, it becomes polar. • Hydroxide is negatively charged, therefore it is attracted to the partial positive charge on a carbon. IB HL Objective • 20.2.2 Describe and explain how the rate of nucleophilic substitution in halogenoalkanes by the hydroxide ion depends on the identity of the halogen. • 20.2.3 Describe and explain how the rate of nucleophilic substitution in halogenoalkanes by the hydroxide ion depends on whether the halogenoalkane is primary, secondary or tertiary. Objectives 20.2.2 & 20.2.3 • One group look at the polarity differences between different halogens and carbon bonds. Predict which halogen would most likely react. • One group look at the primary, secondary and tertiary structure with one type of halogen, and describe how the polarity would differ. Predict the mechanism for each and which would react fastest. • When time is up, you will share your findings with the other group. Objectives 20.2.2 & 20.2.3 • • • • • Summary (Different from what you predicted?) Polarity of the molecule is not as important as the strength of the carbon-halogen bond. Iodine is the least polar, but the most reactive, since the carbon-iodine bond is the weakest. Reactivity of halogenoalkanes depends on the ability of the halogen bond to break and leave, which is why the halogen is referred to as the leaving group. Iodine is the best leaving group, and fluorine is the worst. You learn best from your mistakes. Objectives 20.2.2 & 20.2.3 • Primary halogenoalkanes proceed by an SN2 mechanism. • Tertiary halogenoalkanes proceed by an SN1 mechanism. • Secondary halogenoalkanes can proceed by either SN1 or SN2. • Experimentally, the SN1 reactions are found to be faster than the SN2 reactions. • This could be due to the activation energy being less to form a carbocation then the activation energy needed to form the transition state (five bonds) for SN2. Objectives 20.2.2 & 20.2.3 • • • • Review What would be more likely to react, 1fluorobutane or 1-bromobutane, and why? What is the order, in increasing rate of reaction, for the primary, secondary, and tertiary halogenoalkanes? A: 1-bromobutane is more likely to react because of the lower strength of the bromine-carbon bond. A: primary>secondary>tertiary IB HL Objective • 20.2.4 Describe, using equations, the substitution reactions of halogenoalkanes with ammonia and potassium cyanide. • 20.2.5 Explain the reactions of primary halogenoalkanes with ammonia and potassium cyanide in terms of the SN2 mechanism. Objectives 20.2.4 & 20.2.5 • Reaction with ammonia forms a primary amine. • Reaction with the cyanide ion forms a nitrile. • Group 1: Show iodoethane reacting with ammonia. State the mechanism and show transfer of electrons using curly arrows. Name the new molecule. • Group 2: Show iodoethane reacting with a cyanide ion. State the mechanism and show the transfer of electrons using curly arrows. Name the new molecule. Objectives 20.2.4 & 20.2.5 H H C H I C H H SN2 Mechanism + NH3 H C H H H C H NH2 Ethanamine or ethylamine I- cyanide ion with iodoethane (SN2) H CH3 + C H I CH3 H CN CN H - I - propanenitrile SN2 S (substitution) C Notice how we increased the length of the carbon chain. It went from an ethane to a propane. N(nucleophilic) 2(species reacting in the slowest step) IB HL Objective • 20.2.6 Describe, using equations, the reduction of nitriles using hydrogen and a nickel catalyst. 20.2.6 Describe, using equations, the reduction of nitriles using hydrogen and a nickel catalyst. • Due to lengthening the carbon chain with the cyanide ion, we can now make an amine with an extra carbon by reacting with hydrogen and a nickel catalyst: H2/Ni CH3CH2CN → CH3CH2CH2NH2 IB HL Objective • 20.6.1 Describe stereoisomers as compounds with the same structural formula but with different arrangements of atoms in space. 20.6.1 Describe stereoisomers as compounds with the same structural formula but with different arrangements of atoms in space. Isomers Structural (Different Bonding) Stereo (Different shape) Stereoisomers The order in which they are bonded is the same (same structural formula), but they have a different arrangement. 20.6.1 Describe stereoisomers as compounds with the same structural formula but with different arrangements of atoms in space. • Two different types of stereoisomers…. • Geometrical isomers • Optical isomers (enantiomers) Geometrical isomers occur when bonds are unable to rotate freely, also known as restricted rotation. Optical isomers occur when there are four different atoms or groups attached to a single carbon atom. These molecules are often called asymmetric or chiral. IB HL Objective • 20.6.2 Describe and explain geometrical isomerism in non-cyclic alkenes. 20.6.2 Describe and explain geometrical isomerism in non-cyclic alkenes. Molecular Models 1. Two people make but-1-ene. 2. Two people make but-2-ene. 3. One person make 1,2-dichloroethene Can you make the two but-1-ene look different no matter which way you turn it? What about the but-2-ene? What are the two differences in the models? 20.6.2 Describe and explain geometrical isomerism in non-cyclic alkenes. • Same side = cis • Opposite side = trans • They exist separately because the double bond cannot be rotated. • One bond is a sigma bond and one is a pi bond. Pi bond is formed by the sideways overlap of the p orbitals on each carbon atom. Rotation would involve breaking this pi bond. IB HL Objective • 20.6.3 Describe and explain geometrical isomerism in C3 and C4 cycloalkanes. • Seunghwan: make a molecular model of cis1,2 dichlorocyclopropane. • Nibras: make a molecular model of trans-1,2dichlorocyclopropane IB HL Objective • 20.6.4 Explain the difference in the physical and chemical properties of geometrical isomers. • Look at study guide for diagrams and explanations as well. 20.6.4 Explain the difference in the physical and chemical properties of geometrical isomers. • Geometric isomers have different physical properties such as polarity, which gives rise to differences in boiling point, melting point, solubility, etc. • For example, the boiling point of cis-1,2-dichloroethene is 60°C, whereas trans-1,2-dichloroethene is 48°C. This is because the cis isomer is more polar. • However, in but-2-ene-1,4-dioic acid, the trans has higher intermolecular hydrogen bonding between different molecules, because of the polarity of carboxylic acid groups. The cis-isomer reacts when heated to lose water and become a cyclic acid anhydride. IB HL Objective • 20.6.5 Describe and explain optical isomerism in simple organic molecules. • Make a model of butan-2-ol • Make a model of 2-bromobutane 20.6.5 Describe and explain optical isomerism in simple organic molecules. Optical Isomers or Enantiomers • If a carbon atom has four different groups attached to it, then there are two different ways in which these groups can be arranged. • This is known as an asymmetric carbon atom, or a chiral centre. IB HL Objective • 20.6.7 Compare the physical and chemical properties of enantiomers. 20.6.7 Compare the physical and chemical properties of enantiomers. • Enantiomers are so similar, there is very little difference in physical and chemical properties. • Chemical differences can arise when enantiomers interact with other optically active substances. • Chemical reactions which produce a chiral carbon often contain equal amounts of the two enatniomers. This is called a racemic mixture. • The only difference in the physical properties is their interaction with polarised light. • Find some examples in your books/study guides of where different enantiomers can cause different effects, and examples of enantiomers that are non-racemic. IB HL Objective • 20.6.5 Outline the use of a polarimeter in distinguishing between optical isomers. 20.6.5 Outline the use of a polarimeter in distinguishing between optical isomers. • Optical isomers rotate the plane of planepolarized light in opposite directions. • A polarimeter can be used to see how enantiomers rotate the plane of planepolarized light. • You need a light, two polarizing lenses, and a tube in between the lenses to hold the sample of the enantiomer. 20.6.5 Outline the use of a polarimeter in distinguishing between optical isomers. • If the analyzer has to be rotated clockwise, the enantiomer is dextrorotatory (D), or dextro for short. From the Latin word meaning “right”. • If the analyzer has to be rotated counterclockwise, then the enantiomer is laevorotatory (L), or levo for short. From the Latin word meaning “left”. • If both enantiomers are present, they cancel each other out, and it appears optically inactive. This indicates a racemic mixture.