Electronic Behavior of Atoms: Energy Levels & Spectra
advertisement

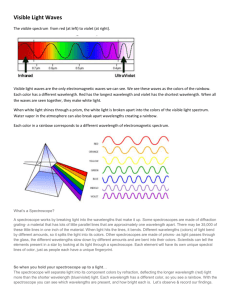
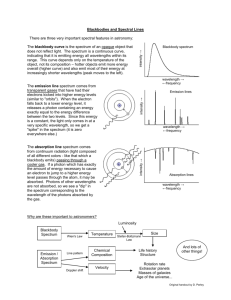
Activity 5: The Electronic Behavior of Atoms Scientist Democritus/Dalton Thomson Rutherford Model of the Atom •Electrons are in fixed energy levels around the nucleus •Energy levels get closer together as they get further from the nucleus. • Think of a ladder that isn’t equally spaced + Where are electrons located? Energy levels: • Regions around the nucleus where electrons can be found •Electrons can move from one energy level to another by gaining or losing energy What’s going on here??? crest trough Short wavelength Long wavelength Wavelength decreases, frequency increases, Energy increases As you move from Red to Violet light, the wavelength of the waves decreases. Long wavelength Low frequency LOW ENERGY Small wavelength High frequency High ENERGY An increase in the frequency of the wave and an increase in energy of the wave. • Colors of light are associated with energy. • Red light has the least energy, while violet light has the most energy. Explain this cartoon….. • Sunlight reaches Earth's atmosphere and is scattered in all directions by all the gases and particles in the air. Blue light is scattered in all directions by the tiny molecules of air in Earth's atmosphere. Blue is scattered more than other colors because it travels as shorter, smaller waves. This is why we see a blue sky most of the time. Why not violet? As the Sun gets lower in the sky, its light is passing through more of the atmosphere to reach you. Even more of the blue light is scattered, allowing the reds and yellows to pass straight through to your eyes. http://news.discovery.com/videos/why-tell-me-whywhy-is-the-sky-blue.html Dan Flavin Flame Test Experiment • Bohr observed the flame colors through an instrument that separates the light into its component colors--just like a prism! Slit (light enters here) Grating diffracts light, separating colors Light source Wavelength scale Look in here This is the spectrum you would observe if you used a spectroscope to look at a white computer screen. How a spectroscope works ATOMIC EMISSION SPECTRUM When the flame colors are observed through a spectroscope, a pattern of bright lines appears ATOMIC EMISSION SPECTRUM The bands of light are called: ATOMIC EMISSION SPECTRUM • All elements have a DIFFERENT pattern of spectral lines that can be used to identify the element. • This spectrum is unique to every element. Why do we see this? • Electrons can get “excited” and move from lower energy levels to higher energy levels by absorbing energy. • When they fall back down to a lower energy level the electron loses energy in the form of light. ATOMIC EMISSION SPECTRUM Atomic Emission Spectrum Add energy So… • Electrons start off in the GROUNDSTATE • They absorb energy and move to the EXCITED STATE • They then lose this absorbed energy fall back down to the groundstate and emit light!