Lecture 6 Review Sheet
advertisement
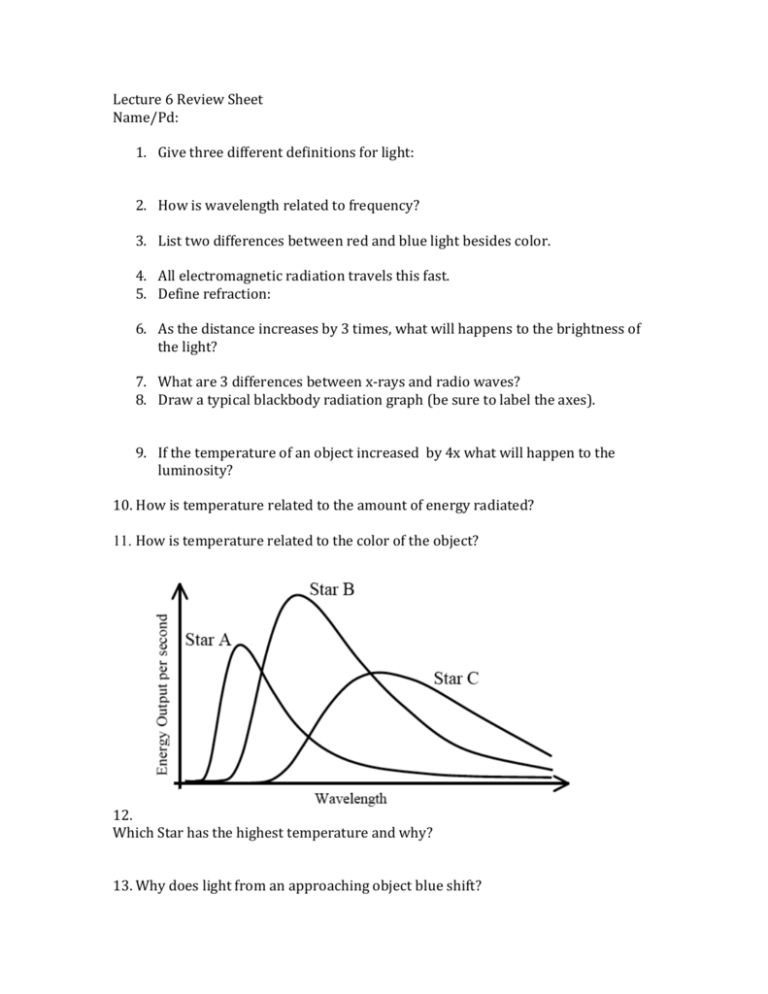
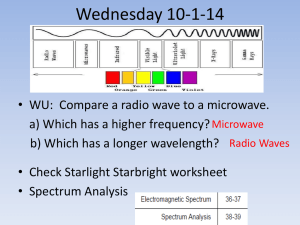
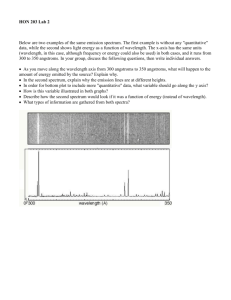
Lecture 6 Review Sheet Name/Pd: 1. Give three different definitions for light: 2. How is wavelength related to frequency? 3. List two differences between red and blue light besides color. 4. All electromagnetic radiation travels this fast. 5. Define refraction: 6. As the distance increases by 3 times, what will happens to the brightness of the light? 7. What are 3 differences between x-rays and radio waves? 8. Draw a typical blackbody radiation graph (be sure to label the axes). 9. If the temperature of an object increased by 4x what will happen to the luminosity? 10. How is temperature related to the amount of energy radiated? 11. How is temperature related to the color of the object? 12. Which Star has the highest temperature and why? 13. Why does light from an approaching object blue shift? 14. What causes the dark lines in an absorption line spectrum? 15. What causes the bright lines in an emission spectrum? 16. Describe how hydrogen, while only having one electron, can have a spectrum with multiple lines. Consider just four of the energy levels in a certain atom, as shown in this diagram: a. Assume an emission spectrum and draw arrows indicating the possible transitions. Hint: which way does the electron jump to release a photon? b. How many spectral lines will result from all possible transitions among these levels? __________ c. Which transition corresponds to the highest frequency (shortest wavelength) light emitted? From n = _______ to n = ________ . d. Which transition corresponds to the lowest frequency (longest wavelength) light emitted? From n = _______ to n = ________ . 18. Give a specific reason (include a number) why you know Barium is not in the unknown composition. 19. Given all things about the same, how should the spectral lines for Hydrogen from Star A compare to that of Star B? 20. Why would a binary star show two slightly different sets of emission lines for hydrogen?