Chapter 3: Chemical Reactions Chapter Review
advertisement

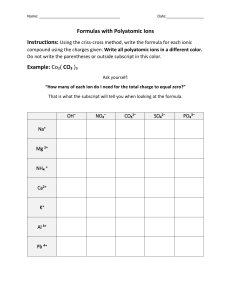
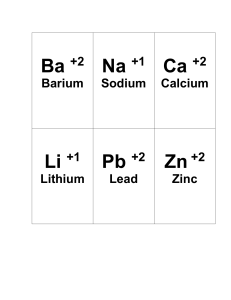
Name _________________________ Period ________ Chapter 3: Chemical Reactions Chapter Review Directions: Write the answer and the page number you found each answer. 1. What is a chemical reaction? 2. What is a chemical change? Give an example. 3. What is a physical change? Give an example. 4. What is a chemical formula? Give an example. 5. What is a chemical equation? Give an example. 6. What are the four evidences of a chemical reaction? Provide an example of each. a. b. c. d. 7. What is a synthesis reaction? Write an example. 8. What is a decomposition reaction? Write an example. 9. What is a combustion reaction? Write an example. 10. What are three physical factors that can be done to speed up a chemical reaction. a. b. c. 11. What is a chemical factor that speeds up a chemical reaction? 12. What is a reactant? 13. What is a product? 14. Copy this equation and circle the reactants and square the product. CH4 + 2O2 CO2 + 2H2O 15. What is a subscript? Give an example of a formula using a subscript. 16. What is a coefficient? Give an example of an equation that uses a subscript. 17. Copy the equation and count the atoms. Is it balanced? 18. Copy the following equation and balance using a T-grid. NH4 + NO3 N2O + H2O 19. What is an endothermic reaction? 20. Where is the energy located? 21. What is an exothermic reaction? 22. Where is the energy located? 23. What is silicon used for in industry? 24. What is respiration?