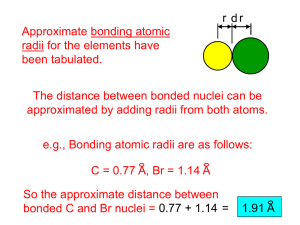
1 GROUP BONDING ALKALI METALS
advertisement

GROUP 1 BONDING ALKALI METALS Bond only by ionic bonding to become 1+ ions with noble gas configurations. The ions are isoelectronic to the noble gas of the PREVIOUS period, which is the kernel of that element. Low electronegativity and low ionization energy causes the outer s electron to be lost. As you read down the group, electronegativity, ionization energy DECREASE, atomic radius INCREASES as does reactivity. See concept map. All positive ions are smaller than the neutral atom from which they are derived, due to a lost electron. EXAMPLES The configuration of Neon 1s2↑↓ The configuration of Sodium (ground) 1s2↑↓ 2s2 ↑↓ 2p6 ↑↓ ↑↓ ↑↓ 2s2 ↑↓ 2p6 ↑↓ ↑↓ ↑↓ 3s1↑_ The configuration of Sodium ION [Na]1+ 1s2↑↓ 2s2 ↑↓ 2p6 ↑↓ ↑↓ ↑↓ + 1e - Ionization and Oxidation The removal of an electron is called IONIZATION, the energy that must be supplied to do so is called IONIZATION ENERGY. This process is endothermic – energy consuming, energy must be supplied and absorbed. In the diagram below, the following is important: 1. The electron removed will most likely be gained by a non-metal. 2. The resulting ion is isoelectronic to the noble gas of the prior period. 3. Ionization energy is needed to remove the electron. Na[Ne] 3s1↑_ 1s2↑↓ 2s2 ↑↓ 2p6 ↑↓ ↑↓ ↑↓ 3s1↑_ Na[Ne]+ + 1e 1s2↑↓ The electron is free of the atom 2s2 ↑↓ 2p6 ↑↓ ↑↓ ↑↓ + 1e-