Document 14452482
advertisement

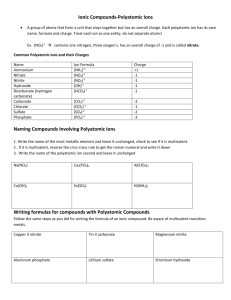
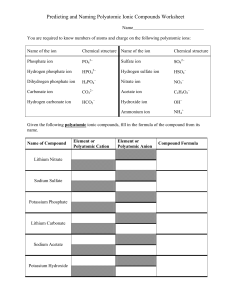
Groups of atoms that tend to stay together and carry an overall ionic charge eg. NO3- SO42- These behave exactly like simple ions to produce a compound with an overall charge of zero Polyatomic Ion Nitrate Formula NO3- Ionic Charge -1 Carbonate Chlorate Sulfate CO32ClO3SO42- -2 -1 -2 Phosphate Hydroxide Ammonium PO43OHNH4+ -3 -1 +1 “Nich Carsulpho” “Nick the Camel ate a Clam Supper in Phoenix” When polyatomic ions are associated with a they form 1. 2. Write the name of the first element (the positively charged ion) Write the name of the polyatomic ion In the case of the polyatomic ammonium, it is written first because it is the positive ion! THE POSITIVE ION ALWAYS COMES FIRST! Name the following compounds: 1. Na2SO4 _______________________ 2. K2CO3 _______________________ 3. NH4NO3 ________________________ 4. Fe3(PO4)2 ________________________ 5. Al(OH) 3 _________________________ 1. Write the symbol of the metal and the polyatomic group. Place brackets around the polyatomic. 2. Write ionic charges for the metal and polyatomic group. 3. Cross over to get the subscript. 4. Simplify, if necessary. (YOU MUST BE CAREFUL NOT TO REDUCE THE POLYATOMIC GROUP!) Example: sodium sulfate is _________ calcium hydroxide is _________ Write the chemical formulas for the following compounds: 1. sodium carbonate _______________________ 2. magnesium nitrate _______________________ 3. sodium hydroxide _______________________ 4. copper(II) sulfate _______________________