Effects of vasoactive agents on isolated human umbilical
advertisement

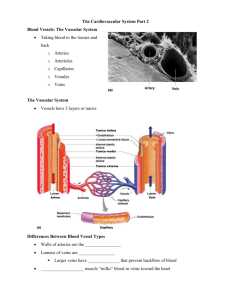
AMERICAN JOURNAL OF PHYSIOLOGY Vol. 222, No. 2, February 1972. Printed in U.S.A. Effects of vasoactive agents on isolated umbilical arteries and veins BURTON M. ALTURA, D. MALAVIYA, CHARLES Departments of Anesthesiology and Physiology, Albert Einstein Bronx, New York 10461 ALTURA, BURTON M., D. MALAVIYA, CHARLES F. REICH, AND Effects of vasoactive agents on isolated human umLOUIS R. ORKIN. bilical arteries and veins. Am. J. Physiol. 222(Z): 345-355. 1972.In vitro experiments, using both helically and longitudinally cut human umbilical arteries and veins (HUAV), were designed to determine: I) whether any of the known circulating vasoactive substances can induce contraction of these vessels in low (physiologic?) concentrations; 2) the relative potency of these substances; 3) the existence of specific drug receptors which subserve contraction; and 4) the relative contribution each type of smooth muscle layer (i.e., longitudinal vs. circular) may play in response to vasoactive agents and in development of spontaneous mechanical activity. The results indicate : 1) 10 different vasoactive agents (amines, polypeptides, prostaglandins, and potassium ions) induce varying degrees of unequal contractile responses (serotonin = maximum); 2) only serotonin ( <O. 1 pg/liter), bradykinin (0.5-4 pg/liter), and oxytocin (l-5 mU /liter) induce contractile responses in very low (physiologic?) concentrations; 3) specific drug receptors appear to exist for all 10 agents in HUAV; 4) dose-response curves for longitudinally cut strips do not always parallel those of the helically cut strips; and 5) circular smooth muscle appears to play a greater role than longitudinal smooth muscle in spontaneous mechanical activity of HUAV. vascular smooth ous activity; drug muscle; bradykinin; umbilical cord; spontanereceptors; neonatal circulation; neurohypophyseal hormones; prostaglandins; serotonin; catecholamines CONTROL OF UMBILICAL CORD BLOOD FLOW IN MAN is, at present, largely speculative ( 14). Although it is well known that the primary factor contributing to the low fetal systemic vascular resistance is the umbilicoplacental circulation (9), the exact site or sites of this resistance is not known. At birth, interruption and eventual obliteration of the umbilical cord vessels take place. The exact mechanism responsible for the latter is, however, not known ( 15). Recently, several investigators have perfused intact umbilical cords and segments of umbilical arteries in vitro in order to assess the pharmacologic behavior of these vessels to various drugs and humoral substances ( 11, 13, 17, 19, 24, 25). But as early as 1938, von Euler (35) noticed that it is extremely dificult, if not impossible, to obtain physiologic flow values in such perfused preparations even if perfused at normal physiologic pressures. In 1965 gtembera and his colleagues (33) measured flow in vivo in the umbilical vein by a local thermodilution technique just after birth and re- human F. REICH, AND LOUIS R. ORKIN College of Medicine of Yeshiva University, ported a mean of 75 ml of blood per minute per kilogram of baby’s weight or approximately 248 ml/min in an average fetus weighing 3.3 kg. Yet the highest reported flows in perfused preparations are 60-80 ml/min (25, 35). Furthermore, in 1967 Gokhale and his co-workers ( 19) working with intact human umbilical cords in vitro, in which only one artery was perfused, reported a tremendous variability from one preparation to another in response to drugs. None of these previous studies with perfused umbilical arteries and veins clearly demonstrates, quantitatively, the relative sensitivity of either human umbilical arteries or veins to vasoactive substances. Attempts in this direction have recently been made by Somlyo et al. (30) using helically cut umbilical artery and vein strip preparations as well as by Eltherington and his colleagues ( 17), using perfused segments and isolated rings of human umbilical artery. Although these latter two studies, when taken together, have demonstrated a high sensitivity of these vessels to serotonin and bradykinin, studies on quantitative dose-response relationships and the existence of specific drug receptors have not been reported in detail. In addition, since, histologically, human umbilical arteries and veins contain two layers of smooth muscle (27, 3 1, 32), an inner one oriented longitudinally, and the outer one circularly, it is impossible to determine from perfused preparations or helical strip preparations alone the relative contribution or role each type of oriented smooth muscle layer may play in response to vasoactive substances. In vitro using both helically and longitudinally cut experiments, human umbilical arteries and veins, were therefore designed to determine: 1) whether any of the known circulating vasoactive substances can induce contraction of these vessels in low (physiologic?) concentrations; 2) the relative potency of these substances; 3) the existence of specific drug receptors which subserve contraction; and 4) the relative contribution each type of smooth muscle layer may play in response to vasoactive agents. METHODS Collection and preparation of human umbilical arteries and veins for in vitro study. A total of 90 human umbilical cords from normal full-term spontaneous deliveries were utilized for this study (Caesarean sections were not utilized). Cords from mothers exhibiting eclampsia, hypertension, diabetes, an Rh factor problem, or other overt diseases were not included 345 346 in this study. Cords from mothers on medications during the last two months of pregnancy, such as antihistamines, morphine, adrenergic blockers, or anticholinergic drugs were also excluded. Fifteen- to 25-cm segments were cut from the cords midway between the placentas and infants as this area is nerve free ( 13, 30, 35, 37). Initially, in our early studies, we immediately flushed the severed cords with KrebsRinger bicarbonate solution, the composition of which has been given previously ( 1, 6), in order to expel the blood. This procedure, however, was soon abandoned for two reasons: I) the cords would expand and bulge under the forced Ringer solution; and 2) we found that this drastically cut down on the reactivity of the tissues to drugs. In subsequent studies we found that if one gently squeezed the cords, in order to expel blood, the preparations would retain a very high sensitivity to drugs. Therefore, this procedure was employed in all studies. The arteries and veins were then carefully dissected out of the cords, using microdissecting instruments. The cord and the dissected umbilical vessels were continuously bathed in cold Krebs-Ringer bicarbonate buffer ( 1, 6). Approximately 385 helical and longitudinal arterial and venous strip preparations were utilized for the present in vitro study. The arteries were cut, helically and longitudinally, 1.3-1.5 mm in width by 25 mm in length while the veins were cut, helically and longitudinally, 2.5-3 mm in width by 25 mm in length. The four types of preparations were then simultaneously set up, isometrically in four separate ZO-ml muscle baths, using procedures and instrumentation previously described elsewhere ( 1, 6), and allowed to equilibrate for 2 hr in Krebs-Ringer bicarbonate under a resting tension of 2 g. The loaded tensions were maintained and periodically adjusted throughout incubation and the experiments. The Krebs-Ringer solutions were oxygenated continuously with a 95 % 02-5 % CO2 mixture and kept at 37 C (pH 7.2-7.4). Drug-induced contractions. Complete cumulative dose-response curves ( 1, 6) as well as single-dose stimulations were obtained on the human umbilical arteries and veins for serotonin, histamine, bradykinin, potassium, prostaglandins, oxytocin, vasopressin, norepinephrine, epinephrine, acetylcholine, and angiotensin in the presence and absence of specific drug antagonists. Each vascular strip was stimulated to contract by serotonin (in maximal or supramaximal concentrations or complete dose-response curves) periodically throughout the experiments since this vasoactive substance induces the greatest maximum response in isolated human umbilical arterial and venous strip preparations (see RESULTS). All drugs and vasoactive substances were randomized in the experiments, i.e., all of the drugs, including serotonin, were added to the isolated organ baths in a different sequence from one preparation to another. In other experiments, paired longitudinal and helical venous and arterial umbilical strips were placed in preweighed Pyrex beakers for dry (oven at 110 C for 18-24 hr) tissue weight determinations. Drugs and chemicals. The chemicals used to make up the Krebs-Ringer bicarbonate solutions, as well as the potassium chloride (KCl), were all American Chemical Society certified reagent grade. The following vasoactive agents and drug antagonists were utilized : epinephrine (Adrenalin chloride, Parke, Davis & Co.), norepinephrine (Levophed ALTURA, MALAVIYA, REICH, AND ORKIN Bitartrate, Winthrop Laboratories), histamine (histamine dihydrochloride, Nutritional Biochemicals Corp.), serotonin (5-HT) (serotonin creatinine sulfate, Nutritional Biochemicals Corp.), angiotensin (angiotensin amide, Hypertensin, Ciba Pharmaceuticals), acetylcholine (acetylcholine chloride, Nutritional Biochemicals Corp.), bradykinin (BRS 640, Sandoz Pharmaceuticals), prostaglandin FZa, prostaglandin Al, oxytocin (synthetic oxytocin, preservative free, 400 IU/ ml, Sandoz Pharmaceuticals), vasopressin (synthetic 8arginine vasopressin, approx. 450 IU/mg, Sandoz Pharmaceuticals), phenoxybenzamine (Dibenzyline hydrochloride, Smith, Kline & French Laboratories), atropine (atropine sulfate, Mann Research Laboratories), UML-49 1 (Methysergide, Sandoz Pharmaceuticals), and pyrilamine (pyrilamine maleate, K and K Laboratories). All of the vasoactive drug stimulants and pharmacologic antagonists, except for the prostaglandins, were made up fresh in triply distilled, deionized water, and concentrated stock solutions so that the total volumes added to the ZO-ml muscle baths never exceeded 1.0 ml. The prostaglandins were dissolved in a solution of 0.095 % ethyl alcohol and 0.00 18 % sodium carbonate in triply distilled, deionized water. All of the vasoactive drug concentrations (or doses) expressed below are in terms of the salt, except for angiotensin, bradykinin, prostaglandins, oxytocin, and vasopressin which are given a .sfree bases. RESULTS S’ontaneous mechanical activity of human artery and venous str$s in vitro. Initially upon in vitro incubation (i.e., the first Z-10 min), the umbilical arterial and venous strips contract from 20 to 60 % over the base-line resting tension of 2 g. Thereafter (i.e., usually within 20-40 min), 90 % of the venous helical and longitudinal strips developed spontaneous mechanical activity. Only 20-30 % of the corresponding artetial preparations would, however, develop spontaneous mechanical activity, and this usually occurred 20-30 min after the veins exhibited signs of spontaneous contractions, and this in turn usually lasted for only 1-2 hr while the spontaneous rhythmic mechanical activity seen in the veins usually persisted, to different degrees, for 5-10 hr. Figure 1 demonstrates the typical spontaneous contractile activity patterns seen in paired (taken from same umbilical cord) helical and longitudinal umbilical arteries and veins. Two things become apparent from such tracings: I) the amplitudes of the helical preparations are at least 2-4 times larger than their corresponding longitudinal preparations; and 2) 4 / HELICAL ARTERY II, LONGITUDINAL HELICAL ARTERY VEIN min bml d LONGITUDINAL FIG. 1. Spontaneous contractile activity in helically dinally cut human isolated umbilical arteries and veins. are taken from same umbilical cord. VEIN and longi tuAll 4 tracings REACTIVITY OF HUMAN UMBILICAL 347 VESSELS the frequency of spontaneous contractions in the longitudinal preparations is greater than in their corresponding helical preparations. Occasionally both the longitudinal and helical arteries and veins exhibited single, spontaneous contractions which, in magnitude, were 70-85 % of a serotonininduced maximal response. This, however, did not occur during the first 2 hr of incubation. This phenomenon could, however, be induced in most preparations by quick stretch or addition of cold Krebs-Ringer bicarbonate solution. Reproducibility of cumulative dose-response curues to 5HT. In 1962 Panigelc (25) observed that perfused umbilical vessels exhibited a tremendous variability in response to serotonin, but which was clearly not tachyphylaxis. In view of these findings, we thought it advisable to determine the reproducibility of helically and longitudinally cut vessels to repetitive serotonin stimulation. Figure 2 demonstrates that although there is variability in response to serotonin, as noted by the large bars representing the standard errors of the means, the cumulative dose-response curves on helically cut umbilical arteries are not significantly different from one another over a period of 8-9 hr. Similar observations were noted on the corresponding longitudinally cut arteries. Figure 3, however, demonstrates that the maximum 5-HT responses on the veins during the first 3.5 hr were less than 40 35 N-10 3c 1o-9 FIG. 2. duced by lative log that were 1o-8 SEROTON lo-’ IN (Molar 1o-6 1o-5 cont.) Effects of incubation in Krebs-Ringer on contractions inserotonin in helically cut human umbilical arteries. Cumudose-isometric response curves. N = number of preparations obtained from different umbilical cords. Brackets = SEM. those seen during the subsequent stimulations. This, however, is not unusual since incubation for 2-4 hr is usually required for excised blood vessels to fully recover from surgical procedures (5). It is of interest to note that serotonin in a concentration as little as lo-lo M can induce threshold contractile responses on these umbilical vessels (Fig. 2). Relative sensitivity of human umbilical arteries and veins to uasoactiue substances. Figures 4-7 demonstrate that of all the vasoactive substances examined for contractile properties on helically and longitudinally cut umbilical arteries and veins serotonin exhibits the greatest relative sensitivitv as well as potency in that it elicits the maximal contractile response. Although all of the vasoactive substances that we have examined can induce contractile responses on these vessels, they produce different degrees of unequal maximal contractile responses (Figs. 4-7). Although there are slight quantitative differences in threshold concentrations, ED50 dose levels and maximal responses, the relative sensitivity of the paired arterial and venous preparations to vasoactive substances is similar. In addition, it should be noted that: I) not every arterial or venous strip would always respond to acetylcholine, angiotensin, epinephrine, norepinephrine, and vasopressin ; and 2) these latter substances in many cases would not elicit graded, cumulative dose-dependent contractile responses; that is to say, these drugs produced all-or-none type contractile responses in some arterial and venous strip preparations. For example, 4 out of 41 arteries and 7 out of 26 veins responded in an all-or-none manner to acetylcholine. Of the five agents capable of evoking this type of response, angiotensin had the highest frequency of all-or-none type responses, occurring in approximate1 .y 50 % of the arterial and venous strips that we examined. Vasopressin produced all-or-none-type contractile responses in 10 of 22 vein preparations. Drug-induced unequal maximal responses. Since the above cumulative dose-response data suggested that all of the vasoactive drugs can induce different degrees of unequal maximal contractile responses, it was of interest to determine whether single supramaximal doses of these vasoactive substances would yield similar unequal maximal responses. Figures 8 and 9 show that such unequal maximal contractile responses are, indeed, obtained on helical and longitudinal arterial strips, using supramaximal drug doses. It is interesting to note that serotonin, as well as the other drugs, quantitatively induces comparable maximal contractile responses on both types of arterial preparations. Although unequal maximal contractile responses are also obtained on helically and longitudinally cut umbilical veins using supramaximal drug doses, serotonin, as well as most of the other drugs, induces, quantitatively, a significantly contractile re(P < 0.01, Student t test) greater maximal sponse on the helical umbilical vein when compared to the longitudinal vein (Fig. 9). However, norepinephrine is approximately equal in magnitude while oxytocin, vasopressin, and angio tensin developed greater tensions on the longitudinally cut venous strips. Serotonin-induced contractile responses on helical and longitudinal umbilical strips. When a comparison of the dose-response relationships for serotonin is made for all four types of umbilical preparations, it becomes apparent that the dose- ALTURA, MALAVIYA, REICH, AND ORKIN TT T T T FIG. 3. Effects of incubation in KrebsRinger on contractions induced by serotonin in longitudinally cut human umbilical veins. Cumulative log doseisometric response curves. N = number of preparations that were obtained from different umbilical cords. Brackets = SEM. lo-lo 1O-9 lo+ SEROTONI 1O-7 N (MOLAR 10+ CONC.) 1O-5 1o-4 . -. HISTAMINE (23) / 10-10 10-9 10 I 10-S -6 AGONIST (Molar 10-4 10-3 10-2 10" cont.) FIG. 4. Relative sensitivity of helically cut human umbilical arteries to vasoactive substances. Cumulative log dose-isometric response curves. Brackets = SEM. All data are presented in terms of percent 5-HT maximal responses. 100% response to 5-HT = 3.42 g ZJZ 0.32 SEM. Numbers in parentheses denote number of different vascular strips, obtained from different cords, utilized response curve. ANG = angiotensin; VP NE = norepinephrine; OXY = oxytocin; Al ; PGFz, = prostaglandin Ffa! ; and ACH response relationships for the helical and longitudinal arteries as well as the helical vein are almost identical, while the dose-response relationship for the corresponding longitudinal vein is distinctly different (Fig. 10). Neurohypophyseal hormone action on human umbilical arteries and veins. The phenomenon illustrated in Fig. 10 is not related exclusively to serotonin-induced contractions. A similar dissimilarity-in dose-response relationships is illustrated in Fig. 11 for two other vasoactive materials, namely oxytocin and vasopressin, but with one important difference. This time the helical umbilical veins, instead of the longitudinal veins, show a different dose-response relationship for oxytocin and vasopressin. Another point of interest shown in Fig. 11 is the relatively greater affinity of oxytocin over vasopressin for the neurohypophyseal peptide receptor in human umbilical arteries and veins. It should be noted that oxytocin and vasopressin were not tested together on the same preparation since we found early in our studies that these substances exhibited profound cross tachyphylaxis, similar to that reported previously by others (30). Speczjkty of drug receptors which subserve contraction in human umbilical arteries and veins. Figures 12-14, when taken together with the data summarized in Table 1, reveal that four different types of pharmacologic antagonists, namely an antihistamine, pyrilamine; an a-adrenergic blocking drug, phenoxybenzamine; an anticholinergic blocker, atropine; for each cumulative dose = arginine vasopressin; PGAl = prostaglandin = acetylcholine. REACTIVITY OF HUMAN UMBILICAL VESSELS 349 AGONIST 5. Relative sensitivity of longitudinally arteries to vasoactive substances. Cumulative sponse curves. All data are presented in terms FIG. human umbilical log dose-isometric reof percent 5-HT maxicut (Molar cont.) ma1 responses. 100% response to 5-HT = 3.21 g rf= 0.36 SEM. EPI = epinephrine. All other symbols and conventions are similar to those used in Fig. 4. HISTAMINE (7) KCI (IO) I 10-10 i 10’9 10-8 10-7 10-b AGONIST 10-S (MOLAR CONC I 10-4 f 10-X 10-2 I lo-’ ) FIG. 6. Relative sensitivity of helically cut human umbilical veins to vasoactive substances. Cumulative log dose-isometric response curves. All data are presented in terms of percent 5-HT maximal responses. 100% response symbols and conventions to 5-HT are similar = 3.30 g zt 0.85 SEM. All to those used in Fig. 4. other and the antiserotonin, UML-491, rather specifically and selectively antagonize histamine, norepinephrine, acetylcholine, and serotonin-induced contractions, respectively, in these isolated blood vessels. Contribution of tissue mass to drug-induced unequal maximal 2 indicate that contractile responses. The data in Table although there might be a trend for the longitudinally cut arteries and veins to be somewhat larger in total mass than the corresponding helically cut arteries and veins, the total dry tissue weights for the paired umbilical arteries and veins are not statistically different from one another. It is apparent from these results, and the developed, maximal isometric tensions shown in Figs. 8 and 9, that a simple relationship between mass of smooth muscle cells1 and the contractile responses to the various drugs cannot account completely for the relative differences in magnitudes of unequal maximal contractile responses in longitudinally vs. helically cut strips. DISCUSSION The present findings which of the spontaneous mechanical indicate activity 1 We have used the dry tissue weight mass of smooth muscle cells responsible metric tension. that the magnitudes found in helically cut as a rough indicator for the development of the of iso- 350 ALTURA, 1o-lO 1o-9 - lo-8 lo-7 - lo-6 AGON FIG. 7. Relative sensitivity of longitudinally veins to vasoactive substances. Cumulative sponse curves. All data are presented in HELICAL cut human umbilical log dose-isometric reterms of percent 5-HT ~~~ LONGITUDINAL 5-HT- SEROTONIN (N-43) H - HISTAMINE (N-23) BK- BRADYKININ (N- 8) Fza - PROSTACLANDIN F2a (N- 5) A, - PROSTAGLANDIN A, IN- 5) K - POTASSIUM CHLORIDE(N- 24) AN6 - ANCIOTENSINIi (N- I I) NE - NOREPINEPHRINE (N- 18) ACH- ACETYLCHOLINE (N-21) OXY- OXYTOCIN (W- 12) VP - VASOPRESSIN IN- 12) T 5-HT (N=36) F20 (N= 5) H (N=21) A, (N= 5) BK (N= 7) K (N=24) OXY (N= 9) ACH (N= 19) NE (N= 14) ANG(N= 8) VP (N= 11) T 5-tiTFza H A, BK K OXYACHNEANCVP HELICALLY CUT STRIPS LONGITUDINALLY CUT STRIPS 8. Unequal maximal contractile responses of various agonists on isolated human umbilical arteries. Vertical bars with brackets represent mean isometric response =I= SEM induced by supramaximal doses of various vasoactive substances (5-HT = serotonin, 5 lug/n-J; H = histamine, 40 &ml; BK = bradykinin, 5 ,ug/ml; Feat = prostaglandin F 2a, 20 pg/n-d;& A1 20 &ml; K = po= prostaglandin tassium chloride, 80 mu; ANG = angiotensin, 5 pg/rnl; NE = norepinephrine, 20 pg/rn.l; ACH = acetylcholine, 40 pg/m.l; OXY = oxytocin, 0.1 U/ml; and VP = arginine vasopressin, 0.2 U/ml). of different cords utilized. N = number FIG. 10-S I ST (Molar maximal All other MALAVIYA, lo-4 REICH, AND ORKIN ‘0” cont.) responses. 100% response to 5-HT symbols and conventions are similar = 1.80 g =t 0.25 SEM. to those used in Fig. 4. umbilical arteries and veins are much greater than their corresponding longitudinally cut counterparts can be correlated to previous histological findings which indicate that the outer layer of circularly arranged smooth muscle cells, in the walls of these human umbilical vessels, is much larger in muscle mass than the inner longitudinal layers (3 1, 32). The rhythmical behavior of these helically and longitudinally cut arteries and veins suggests that: I) cell-to-cell conduction in both layers of smooth muscle is probably quite good; and 2) there are probably numerous regions of close membrane apposition or numerous nexus areas which function as low-resistance pathways ( 10). The finding that the frequencies of spontaneous activity in the longitudinally prepared umbilical vessels are slightly greater than their corresponding helically prepared counterparts is suggestive of more rapid cell-to-cell conduction in the longitudinal than in the latter circular smooth muscle cell layers. One could also entertain the possibility that conduction in the circular smooth muscle layers is naturally slower and may be functionally important in the in vivo situation. Electronmicrographic and electrophysiological studies should throw considerable light on the characterization of the individual smooth muscle layers. Although it is always difficult, if not dangerous, to extrapolate in vitro findings to the in vivo situation, one must entertain the possibility that these strong (as great as 75-80 % maximum response in certain cases), spontaneous, and rhythmic contractions of the individual layers of smooth muscle cells may play an important role in propelling blood to and away from the fetus in utero. The data presented here indicate that a variety of vasoactive substances (amines, polypeptides, prostaglandins, and potassium ions) induce unequal maximal contractile responses in human umbilical arteries and veins, similar to that noted previously for various somatic arteries and veins in animals ( 1, 4, 6, 29). The present quantitative in vitro data, employing a wide variety of vasoactive agonists, suggest that the relative number of potential drug receptors or REACTIVITY OF HUMAN UMBILICAL 351 VESSELS 4.c HELICAL 5-HT= H = BK = K = ACH= NE = OXY= VP= ANG= LONGITUDINAL SEROTONIN (N=18) HISTAMINE (N= 7) BRADYKININ (N= 5) POTASSIUM (N=lO) ACETYLCHOLINE (N=9) NOREPINEPHRINE (N =8) OXYTOCIN (N= 5) VASOPRESSIN (N=6) ANGIOTENSIN (N=5) 5-HT(N=37) H (N=20) BK(N4 1) ACH(N= 17) K(N=18) NE(N=15) OXY(N= 17) VP(N= 5) ANG(N= 6) 3.5 (42) (20) 3.0 (36) 25 xii $y, 2.0 z Lu b (36) : g 1.5 s1 - 1.0 0.5 0 ,,L_,o HELICALLY CUT STRIPS LONGITUDINALLY - 10-9 I I I 10-7 10-6 1o-5 SEROTONIN CUT STRIPS FIG. 9. Unequal maximal contractile responses of various agonists on isolated human umbilical veins. Symbols and conventions are similar to those used in Fig. 8. Supramaximal drug doses were utilized (5-HT = 20 pg/ml; H = 80 pg/ml; BK = 5 ,ug/ml; K = 80 mM; ACH = 40 pg/ml; NE = 20 pg/ml; OXY = 0.1 U/ml; VP = 0.2 U/ml; and ANG = 5 pg/ml). N = number of different cords utilized. I 10-8 (Molar cod 10. Comparison of contractile responses induced by 5-HT on isolated longitudinally and helically cut human umbilical arteries and veins. Cumulative log dose-isometric response curves. HUA = helical umbilical artery; HUV = helical umbilical vein; LUA = longitudinal umbilical artery; and LUV = longitudinal umbilical vein. Numbers in parentheses denote the number of different cords utilized. Values are means 3~ SEM. FIG. T T A-OXY (12) LUA TT 5 0.8FIG. 11. Neurohypophyseal hormone action on isolated longitudinally and helically cut human umbilical arteries and veins. Cumulative log doseisometric response curves. OXY = oxytocin; VP = arginine vasopressin. Other symbols and conventions are similar to those used in Fig. 10. Values are means is z Z :" z Oh- 5 0 2 0.4- =t: SEM. --OXY i NEUROHYPOPHYSEAL HORMONE (U/L) \ \ A -VP(7) (IO) HUV HUV ALTURA, 352 t t t 5-HT H PY. BK + H + 5-HT PY + H + H OiY PY BK H + 5-HT PY + H b OXY PY + H t BK .1, min I I I ~~ CONTROLS EXPERIMENTALS t t t NE 5-HT t t Ach BK PBZ ~~ -1 PBZ + NE + Ach PBZ + BK mm ARTERY t t KCIt NE t 5-HT PBZ Ach + NE + 5-HT PBZ +NE + KCI PBZ + Ach t-J-4 mln CONTROLS EXPERIMENTALS AND ORKIN (PYRILAMINE) t tt t tt t NE +5-HT + REICH, FIG. 12. Influence of pyrilamine on drug-induced contractions in longitudinally cut human umbilical vein and artery. Note that preincubation (15 min) with the antihistamine pyrilamine (0.5 pg/ml) completely blocks contractions induced by histamine (H) (20 &ml) but does not affect contractions induced by 5-HT (0.5 pg/ml), bradykinin (BK) (0.5 pg/ml), or oxytocin (OXY) (0.05 U/ml). Antihistamine remained in bath in contact with vascular strips during addition of drug agonists. BK ARTERY 5-HT MALAVIYA, FIG. 13. Influence of phenoxybenzamine on drug-induced contractions in helically cut human umbilical vein and artery. Note that preincubation (15 rnin) with a+adrenergic antagonist phenoxybenzamine (0.05 pg/ml) completely blocks contractions induced by norepinephrine (NE) (1.0 pg/ml) but does not affect contractions induced by 5-HT (0.5 pg/rn.l), acetylcholine (Ach) (10 pg/ml), bradykinin (BK) (0.5 pg/ ml), or KC1 (40 mM). Phenoxybenzamine remained in bath in contact with vascular strips during addition of drug agonists. (PHENOXYBENZAMINE) VEIN t 5-HT t t J-i H Ach AT + A;h min + S-HT AT + Ach + H FIG. 14. Influence of atropine on drug-induced contractions in longitudinally cut human umbilical vein and artery. Note that preincubation (15 min) with cholinergic antagonist atropine completely blocks contractions induced by acetylcholine (Ach) (10 pg/ml) but does not affect contractions induced by 5-HT (0.5 lug/ml) or histamine (H) (20 pg/rnl). Atropine remained in bath in contact with vascular strips during addition of drug agonists. ARTERY lgmC t Lt t --,+I t t t -tt f t 5-HT H Ach AT + Ach A min + S-HT AT + Ach + H I I 1 I I CONTROLS i EXPERIMENTALS (ATROPINE) reactive smooth muscle cells for different drugs is not necessarily the same for the longitudinal and circular smooth muscle layers, even though the maximal responses for a parFor titular agonist may be quantitatively equivalent. example, serotonin elicits comparable degrees of isometric tension development in longitudinal and helical arterial preparations (Fig. 8), but the relative decreasing order of unequal maximal contractile responses elicited by the other vasoactive substances is not the same. Furthermore, previous histological studies (3 1, 32) indicate that the outer circular layer of smooth muscle cells in both the human umbilical artery and vein is many times thicker (and thus much richer in total numbers of smooth muscle cells) than the inner longitudinal smooth muscle layer, yet: a) the maximal isometric REACTIVITY TABLE OF HUMAN UMBILICAL VESSELS 1. Influence of various pharmacologic 353 antagonists on drug-induced contractions in human umbilical arteries and veins Agonist Antagonist p Ax, Ach*, 10 ccg/ml Pyrilamine, 0.5 pg/ ml Phenoxybenzamine, 0.05 j&ml Atropine, 0.5 pg/ml UML-491, 0.5 Irg/ml - t w (4) + w - 0.05 /%/ml > (3) * Ach = acetylcholine; Vp OXY = oxytocin; $ Number of different to respond to agonist Ang = 8-arginine preparations in presence = 0.5 tk/rnl (3) (37.5) (4) - (5) - (2) (3) angiotensin vasopressin; (arteries of antagonist. EpL Hist, 20 pg/ml 1.0 erg/ml (3) + w- > (3) (2) - (8) (3) - amide; Bk = bradykinin; PGAr = prostaglandin and veins). 8 Number TABLE 2. Dry tissue weights of helically and longitudinally cut human umbilical arteries and veins Helical Artery Weight* 6.31 =I= 0.54t V)$ Longitudinal Artery 7.18 & (7) Helical Vein 0.355.39 =t Longiizinal 1.17t6.47 (7) See METHODS for dimensions of various preparations. values (mg * s~hi). t Not significantly different sponding longitudinal artery and vein (P > 0.05). different preparations from different cords. =t 0.48 (7) * Mean from corre$ Number of tensions (i. e., induced by 5-HT) obtained in the present experiments, in the paired longitudinally and helically cut human umbilical vessels are either approximately equal (e. g., arteries) (Fig. 8) or quite different in magnitude (e. g., veins) (Fig. 9); and b) the total masses of smooth muscle cells, on the basis of dry weight determinations, in the paired umbilical arterial and venous strip preparations are not statistically different from one another. It would, therefore, be very difficult to account for the differences in developed tensions in the longitudinal vs. helical preparations solely on the basis of the amount of contractile substance present in the longitudinal vs. circular layers. Although this suggests that heterogeneity of drug receptors exists in the circular and longitudinal smooth muscle layers, the various individual drugs activate specific receptors since a variety of pharmacologic antagonists rather selectively inhibited only the specific drug agonists they were designed to counteract. Serotonin and bradykinin appear to exhibit the greatest affinity for their respective receptors in these vessels; on a molar basis, serotonin is more potent than bradykinin on the umbilical arteries as well as veins. Although these findings, in relation to potency, are the reverse of those found by Eltherington et al. ( 17) for these same two substances on human umbilical arteries, one must keep in mind that these latter investigators used perfused arterial segments. Our isolated helical and longitudinal arterial strip preparations appear to be 6-30 times more sensitive to serotonin than are the perfused arterial segments in the experiments of Eltherington et al. It is of some interest to note that the levels of bradykinin needed to elicit threshold contractile responses (0.5-4.0 rig/ml) are in the range of maternal plas- 0% KCl, 40InM (8) (6) (4) (5) Epi = Al ; 5-HT of different 1 .OT:)rnl 0.05 U/ml VP9 0.05 U/ml - - - (5) + (8) - (f-0 - (3) - (3) - (3) - (4) - @/8 > (3-4) (5) epinephrine; = serotonin. preparations (4) (4) PGAI SHT, 0.5 rg/ml , 10 pgg/ml (3) (2) (3) (3) , / 11 = histamine; NE = norepinephrine; t Minus = no effect;plus = blockade. out of total number examined that failed Hist ma bradykinin concentrations (22). But probably, more importantly, the umbilical cord venous and arterial plasma kinin concentrations found by Melmon et al. (22) at birth were significantly higher than those found in maternal venous plasma, reaching levels 45 times those of adult maternal venous plasma. An extrapolation of these umbilical venous and arterial plasma kinin levels (22) to our work would result in contractile responses which are equivalent to 25-60 % of a serotonin-induced maximal response. Although, to our knowledge, umbilical cord plasma serotonin levels are not known, it should be noted that: I) an increase of 5-HT metabolism takes place in the last months of pregnancy ( 18); and 2) recent findings of Yuwiler et al. (28, 36) indicate that, at least for whole blood, young children have significantly higher serotonin blood levels than do adults (mean values: 0.241 =t 0.053 vs. 0.161 & 0.047 pg/ml). It would not be too unreasonable to suggest that the 5HT levels in umbilical cord plasma may even be higher since previous findings of others have shown that the 5-HT content of the rat fetus increases from undetectable amounts early in pregnancy to 0.2 pg/g at term (16), which might suggest a feedback of 5-HT into the umbilical cord vessels. But even if one considers the blood level of Yuwiler et al. (i. e., 0.241 pg/ml) and extrapolates this to our experiments, this serotonin level (if it is mainly free, unbound 5-HT) could effect contractile responses which are 85-95 % of the maximum responses observed in the present experiments. Furthermore, even if the actual umbilical cord plasma levels of free 5-HT were only 10 % of 0.241 pg/ml, they would result in contractile responses which are 65-80% of the maximum responses observed in the present experiments. At the very least, the possibility must be entertained that bradykinin and serotonin may be of importance in regulation of fetal systemic vascular resistance via a tonic constrictor action on the umbilical cord vessels. Although this study demonstrates that histamine and two prostaglandins (A, and Fgar) can produce near maximal contractile responses in isolated human umbilical arteries and veins, their possible physiologic role seems to be highly questionable, if not precluded, in view of our data which indicate that comparatively high concentrations of these substances are needed for threshold effects. In both cases, that is for histamine (4.0-5.0 rig/ml) and the prostaglandins 354 (.05-0.5 pg/ml), the effective concentrations necessary for threshold contractile responses are higher than either the known assayed maternal plasma levels of these substances or those found at birth (2 1, 23, 26). Even though actual circulating levels of the prostaglandins are far below those needed for threshold contractile effects, one must take into consideration the recent findings of Karim (20) which indicate that the walls of the umbilical cord vessels may contain concentrations of prostaglandins which, if freed or released during pregnancy and/or birth, could induce umbilical vessels to contract. Our studies indicate that angiotensin, acetylcholine, and the catecholamines can also elicit contractile responses on human umbilical arteries and veins. The physiologic role that these latter substances play in regulating umbilic al cord blood flow in man is probably negligible or minimal for several reasons : I) the in vitro concentrations of these substances which are necessary for even threshold contractile effects are higher than those found in man; 2) only some preparations were found to respond to these substances confirming previous findings of others ( 19, 30, 34); 3) many preparations did not respond in a dose-dependent (graded) fashion but in an all-or-none manner; and 4) these vasoactive substances, in those instances where they can produce graded contractions, can effect only a small portion of the maximal response (see Figs. 4-9). oxytocin and Although the neurohypophyseal hormones vasopressin can effect only a small portion of the serotonininduced maximal contractile responses on human umbilical arteries and veins, the dose-response relationships observed in the present experiments are, nevertheless, important for several reasons and should be discussed here. First, oxytocin induces threshold contractile effects in both human umbilical arteries and veins in very low (physiologic?) concentrations (e. g., l-5 mu/liter), confirming and extending previous findings of Somlyo et al. (30). A recent report by Chard and his co-workers ( 12), using a very sensitive radioimmunoassay technique for oxytocin, indicates that human umbilical cord venous and arterial plasma oxytocin levels (obtained immediately after delivery) are on the average between 24 and 45 mu/liter, concentrations which, on extrapolation, are capable of eliciting 20-30 % of maximal oxytocin contractile responses (see Fig. 11). Second, oxytotin consistently (unlike vasopressin) produced contractions in all of the human umbilical arterial and venous preparations that we examined, and oxytocin always elicited a greater portion of the maximal contractile response when ALTURA, MALAVIYA, REICH, AND ORKIN compared to vasopressin. Third, the dose-response curves for oxytocin are shifted to the left of those for vasopressin on each type of preparation (see Fig. 11). These pharmacologic phenomena exhibited by oxytocin and vasopressin are very different from that seen for either of other large somatic mammalian blood vessels 1,3a, 6a) or the small mammalian microscopic blood vessels (3,6a, 7, and unpublished observations) and strongly suggest that the neurohypophyseal peptide receptor in human umbilical arteries and veins is probably different from that in other mammalian blood vessels. These data could, therefore, be marshaled to support our previous suggestion that the neurohypophyseal receptor may not be identical in all mammalian vascular smooth muscle ( l-3). In view of the data presented here, the possibility must be entertained that oxytocin, serotonin, and bradykinin may play an important role in human umbilical cord blood flow in vivo. Quantitative assay of these and other endogenous vasoactive substances in plasma from women, during various stages of pregnancy, should aid in the clarification of the physiologic role for these substances. Since previous, recent work of others has indicated that kinin (22) as well as oxytocin ( 12) umbilical cord vessel plasma levels are markedly increased at birth, one must consider the distinct possibility that these two polypeptides, acting in concert with increased oxygen tensions ( 15) and serotonin, may effect closure of human umbilical cord vessels at birth. The authors acknowledge the many helpful discussions held with Dr. Bella T. Altura. The authors are deeply indebted to the nursing and resident staffs of the Departments of Anesthesiology and Obstetrics of the Bronx Municipal Hospital Center and the College Hospital of the Albert Einstein College of medicine, and to Miss Gertie Marx, Professor of Anesthesiology, without whose assistance these studies would not have been possible. The authors are also indebted to the late Dr. R. Bircher and Dr. B. Berde, Sandoz Pharmaceuticals, for generously supplying the pure, synthetic neurohypophyseal polypeptides used in this study. The authors also thank Dr. Richard Weiner, New York Medical College and Dr. John Pike, The UpJohn Co., for generously supplying the pure, synthetic prostaglandins used in this study. A preliminary account of portions of this work was presented to the Spring 1971 Meeting of The American Society of Pharmacology and Experimental Therapeutics, Inc., April 14, 197 1, Chicago, Ill. (8). This work was supported by Research Grants HE-12462 and HE11391 from the National Heart and Lung Institute. B. M. Altura is a recipient of Public Health Service Research Career Development Award 5-K3-GM, 38, 603. Received for publication 25 June 197 1. REFERENCES 1. ALTURA, B. M. Significance of amino acid residues in vasopressin on contraction in vascular muscle. Am. J. Physiol. 219: 222-229, 1970. 2. ALTURA, B. M. Influence of magnesium and cysteine on vasopressin-induced contractions in various canine blood vessels. Exfierientia 26 : 1089- 1090, 1970. 3. ALTURA, B. M. Pharmacology of neurohypophyseal hormones and analogs on isolated vascular muscle and in the terminal vascular bed. PTOG. Symp. Physiol. Pharmacol. Vascular Neuroeffector Systems, Interlaken, Switzerland, 1969 p. 274-290, 1971. 3a ALTURA, B. M. Structure-activity relationships of neurohypophyseal polypeptides on different types of isolated mammalian blood vessels. Proc. Symj. Vasoactive PoZypeptides, Florence, Italy, 1970. In press. 4. ALTURA, B. M., AND B. T. ALTURA. Heterogeneity of drug receptors in different segments of rabbit thoracic aorta. European J. Pharmacol. 12 : 44-52, 1970. 5. ALTURA, B. M., AND B. T. ALTURA. Calcium content and force of drug-induced contractions of arterial muscle during recovery in vitro. Proc. SOL Exptl. Biol. Med. 135: 739-744, 1970. 6. ALTIJRA, B. M., AND B. T. ALTIJRA. Differential effects of substrate depletion on drug-induced contractions of rabbit aorta. Am. J. Physiol. 219: 1698-1705, 1970. 6a ALTURA, B. ha., AND R. W. BURTON. Structure-activity relationships of neurohypophyseal hormones on rat arterial and arteriolar smooth muscle: relationship of magnesium to contractil activity (Abstract). Federation Proc. In Press. 7. ALTIJRA, B. M., AND S. G. HERSHEY. Pharmacology of neuro- REACTIVITY 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. OF HUMAN UMBILICAL 355 VESSELS hypophyseal hormones and their synthetic analogues in the terminal vascular bed : structure-activity relationships. AngioZogy 18 : 428-439, 1967. ALTURA, B. M., D. MALAVIYA, AND L. R. ORKIN. Comparative contractile effects of vasoactive substances on isolated human umbilical arteries and veins (Abstract). Federation Proc. 30 : 383, 1971. ASSALI, N. S., G. A. BEKEY, AND L. W. MORRISON. Foetal and neonatal circulation. In: Biology of Gestation, edited by N. S. Assali. New York: Academic, 1968, vol. II, p. 85-86. BARR, L. The ultrastructure and biophysics of smooth muscle. In: The Biological Basis of Medicine, edited by E E. Bittar and N. Bittar. New York: Academic, 1969, vol. 6, p. 96-124. BOR, I., AND W. G. GUNTHEROTH. In vitro response to oxygen of human umbilical arteries and of animal ductus arteriosus. Can. J. Physiol. Pharmacol. 48 : 500-502, 1970. CHARD, T., N. R. H. Boyd, M. L. FORSLING, A. S. MCNEILLY, AND J. LANDON. The development of a radioimmunoassay for oxytocin: the extraction of oxytocin from plasma, and its measurement during parturition in human and goat blood. J. Endocrinol. 48 : 223-234, 1970. DAVIGNON, J., R. R. LORENZ, AND J. T. SHEPHERD. Response of human umbilical artery to changes in transmural pressure. Am. J. Physiol. 209 : 5 l-59, 1965. DAWES, G. S. Foetal and Neonatal Physiology. Chicago: Year Book, 1968, p. 66-78. DAWES, G. S. Foetal and Neonatal Physiology. Chicago: Year Book, p. 160-162, 1968. DIXON, J. B. Histamine, 5-hydroxytryptamine and serum globulins in the foetal and neo-natal rat. J. Physiol., London 147: 144152, 1959. ELTHERINGTON, L. G., J. STOFF, T. HUGHES, AND K. L. MELMON. Constriction of human umbilical arteries. Interaction between oxygen and bradykinin. Circulation Res. 22 : 747-752, 1968. GARATTINI, S., AND L. VALZELLI. Serotonin. Amsterdam: Elsevier, 1965, p. 59. GOKHALE, S. D., 0. D. GULATI, L. V. KELKAR, AND V. V. KELKAR. Effect of some drugs on human umbilical artery in vitro. Brit. J. Pharmacol. 27: 332-346, 1967. KAFUM, S. M. M. The identification of prostaglandins in human umbilical cord. Brit. J. Pharmacol. 29: 230-237, 1967. KARIM, S. M. M. Appearance of prostaglandin Fzat in human blood during labor. Brit. Med. J. 4 : 6 18-62 1, 1968. 22. MELMON, possible 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. 33. 34. 35. 36. 37. K. L., M. J. CLINE, T. HUGHES, AND A. S. NIES. Kinins: mediators of neonatal circulatory changes in man. J. Clin. Invest. 47 : 1295-l 302, 1968. MITCHELL, R. G., AND J. F. PORTER. Histamine and granulocytes in the umbilical cord blood of infants at birth. Brit. J. Pharmacol. 40: 310-316, 1970. PANIGEL, M. Contribution to the physiology of feto-placental J. Physiol., Paris 5 1 : 941-969, 1959. circulation. PANIGEL, M. Placental perfusion experiments. Am. J. Obstet. Gynecol. 84 : 1664-1683, 1962. PORTER, J. F., J. A. YOUNG, AND S. RASHEED. Umbilical cord blood histamine levels in newborn infants with and without Biol. Neonatorum 15 : 300-303, 1970. perinatal anoxia. REITH, E. J., AND M. H. Ross. Atlas of Descriptive Histology. New York: Harper and Row, 1970, p. 198. RITVO, E., A. YUWILER, E. GELLER, S. PLOTKIN, A. MASON, AND K. SAEGER. Maturational changes in blood serotonin levels and platelet counts. Biochem. Med. 5 : 90-96, 197 1. SOMLYO, A. V., R. L. SANDBERG, AND A. P. SOMLYO. Pharmacologically heterogeneous smooth muscle cell distribution in blood vessels. J. Pharmacot. Exjtl. Therap. 149 : 106-l 12, 1965. SOMLYO, A. V., Woo, C., AND A. P. SOMLYO. Responses of nervefree vessels to vasoactive amines and polypeptides. Am. J. Physiol. 208 : 748-753, 1965. SPIVACK, M. The anatomic pecularities of the human umbilical cord and their clinical significance. Am. J. Obstet. Gynecol. 52: 387-401, 1946. STALLABRASS, P. Observations on the muscular coat of the human umbilical blood vessels. J. Obstet. Gynaecol. Brit. Commonzuealth 70: 1042-1043, 1963. STEMBERA, 2. K., J. HODR, AND J. JANDA. Umbilical blood flow in healthy newborn infants during the first minutes after birth. Am. J. Obstet. Gynecol. 91: 568-574, 1965. TAKENAKA, F. Response of vascular strip preparations to noradrenaline and tyramine. Japan. J. Pharmacol. 13 : 274-281, 1963. VON EULER, U. S. Action of adrenaline, acetylcholine and other substances on nerve free vessels (human placenta). J. Physiol., London 93: 129-143, 1938. YUWILER, A., S. PLOTKIN, E. GELER, AND E. R. RITVO. A rapid accurate procedure for the determination of serotonin in whole human blood. Biochem. Med. 3 : 426-431, 1970. ZAITEV, N. D. Development of neural elements in the umbilical cord. Arkhiv. Anat. Gistot. i Embriol. 37 : 81-88, 1959.