University of Babylon /College Of Engineering Electrochemical Engineering Dept.
advertisement

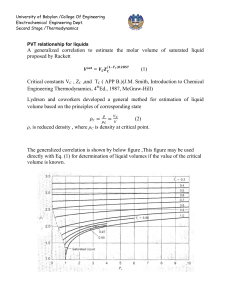
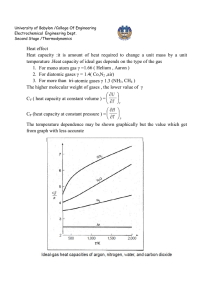
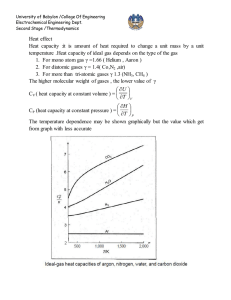
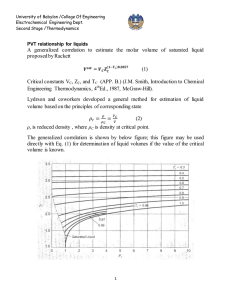
University of Babylon /College Of Engineering Electrochemical Engineering Dept. Second Stage /Thermodynamics Equation of state PV=nRT is the simplest equation of sate and is called ideal gas equation Virial EQUATIONS OF STATE For an isothermal such as T 1 as pressure increase , volume decrease , thus PV product for gas or vapor should be more nearly constant and this lead to suggest that PV along isotherm can be represented as a power series equation , this called Virial equation state PV = 1 + B ¢P + C ¢P 2 + D ¢P 3 + ..... RT PV B C D z = = 1 + + 2 + 3 + .... RT V V V B B¢= RT C -B2 C¢= ( RT ) 2 z = D - 3 BC - 2 B 2 D¢= ( RT ) 3 B,B¢ are second Virial coefficients ,C,C¢ are third Virial coefficients and so on. Values of C, like those of B, depend on the gas and , temperature. The below figure shows a compressibility-factor graph for methane. Values of the compressibility factor Z (as calculated from P VT data for methane by the defining equation Z = P V / RT) are plotted vs. pressure for various constant temperatures. University of Babylon /College Of Engineering Electrochemical Engineering Dept. Second Stage /Thermodynamics · all isotherms originate at value z=1 for P =0. · the isotherms are nearly straight at low pressure . · Thus the equation of the tangent lines is z = 1+ B¢ P dz = B ¢ + 2C ¢P + 3 D ¢P 3 + .... dP æ dz ö = B ¢ = slop ç ÷ è dP ø P =0 \z = PV BP =1+ RT RT ® This equation satisfactorily the PVT behavior of many vapors at subcritical temperature up to pressure of about 5 bar. for pressure the range applicability of Eq. ® but below the critical pressure, the Virial equation truncated to three terms often provides excellent results. ®® PV B C \z = = 1+ + 2 RT V V University of Babylon /College Of Engineering Electrochemical Engineering Dept. Second Stage /Thermodynamics This equation can be solved directly for pressure, but is cubic in volume. Solution for V is easily done by an iterative scheme with a computer. The effect of temperature on C and B for Nitrogen are illustrated on below figure